Hydrogeochemistry and Groundwater Quality Assessment of Rapur Area, Andhra Pradesh, South India ()
Received 11 February 2016; accepted 24 April 2016; published 27 April 2016

1. Introduction
Land and water are critical natural resources that sustain human life and the lives of all other creatures on our planet. Groundwater is one of the primary sources of water for human consumption, agriculture and industrial uses in Rapur area. Determination of physical and chemical quality of water is essential for assessing its suitability for various purposes. Generally, the quality of groundwater depends on the composition of recharge water, the interaction between the water and the soil, the soil-gas interaction, the rock with which it comes into contact in the unsaturated zone, the residence time, and reactions that take place within the aquifer [1] - [5] . Groundwater quality in a region is largely determined by both natural processes (dissolution and precipitation of minerals, groundwater velocity, quality of recharge water, and interaction with other types of water aquifer) and anthropogenic activities [6] . Literature suggests that little work on this aspect has so far been done in India [7] - [9] .
The natural chemical quality of groundwater is generally good, but elevated concentrations of a number of constituents can cause problems for water use. The geochemistry of groundwater data gives crucial evidence to the geologic history of rocks and indications of groundwater recharge, movement, and storage [10] - [12] . Rapid growth of urban population, development of agriculture and industrial activities especially in hard rock terrains caused an intense increase in water consumption. Hydrogeochemical processes are influenced by many factors, such as geogenic factors (i.e., rock-water interaction) and anthropogenic activities (i.e., agricultural, industrial and domestic activities) [3] . Recent research showed the importance of investigating the impacts of various factors on hydrogeochemistry [13] [14] . Groundwater contribution in rural areas for drinking purpose is about 88%, where water treatment and transport do not exist [15] . By understanding the chemistry of groundwater, we can determine its usefulness for domestic and agricultural purposes. If the quality of ground water is good then it can yield better crops under good soil and water management practices. Factors like the quality of water, soil type, salt tolerance characteristics of plants, climate and drainage decides the suitability of irrigation water in agriculture sector [9] [16] [17] . Intensive agricultural activities have increased the demand on groundwater resources in India [18] - [20] . Keeping in view of associated problems with ground water in Rapur area of Nellore District, present study was carried out to find out its suitability for domestic and agriculture purpose.
2. Area of Study
This study area is about 40 sq・km and is located in the Rapur Taluk of Nellore District, Andhra Pradesh. It forms part of the Survey of India toposheet No. 57 N/11 and lies between 14˚16'30''N forms and 14˚19'N latitude and 79˚38'E and 79˚41'E longitude (Figure 1). The area is accessible by the Nellore Rapur road which passes through North-western portion of the area. The general level of cultivated land is about 200 feet from which rise the hillocks of the area namely Guttikonda (689 feet), Varavadikonda and other isolated hillocks (340 feet etc.,). One ridge runs north-south and other runs approximately east-west from the southern end of the former describing as an “L” shaped topographic feature. Along these ridges, the rocks crop above to form isolated small peaks here and there. In spite of the variations in alignment of these two ridges, the rocks that constitute these ridges, maintain uniformity in their strike and dip directions. There are isolated hillocks namely Varavadikonda (478 feet, 344 feet and 312 feet) to the south of Duggenta. These hillocks form the out crops of quartzites. The Guttikonda (687 feet) hillock which is the prominent ridge in this area formed a watershed for few streams and river lets, that flow southwards to form tributaries to the
Pennar
River
. Other river lets flow eastwards and form feeding channels around Duggenta. The area is charactersied by hot and sub-humid climate and is in the tropical region. The climate is, in general, healthy and is not subjected to sudden variations in temperature. The maximum, average and minimum temperatures are 40˚C, 31˚C and 18˚C respectively. They receive rainfall during the months of July, August, September and October with maximum precipitation in October. The heavy rainfall is limited to a few days in a year due to depressions in
Bay of Bengal
which leads to flash floods of high discharge. The average annual rainfall is a little over 1000 mm with high spatial and temporal variations.
3. Materials and Methods
Groundwater samples were collected from 30 locations from Rapur area during April 2014. The collected water samples were transferred into precleaned polythene container for analysis of chemical characters. Samples were analyzed in the laboratory for the 2 physico-chemical attributes like pH, electrical conductivity (EC), total hardness (TH), total dissolved solids (TDS), dissolved silica and major ions (Ca2+, Mg2+, Na+, K+, Cl−, 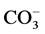 ,
, 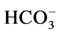 and
and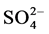 ). All parameters were analyzed by following standard methods [21] . The pH and conductivity were measured by using Systronics micro pH meter model 361 and Deluxe conductivity meter model 601. Total Hardness (TH), Ca2+, Mg2+, Cl−,
). All parameters were analyzed by following standard methods [21] . The pH and conductivity were measured by using Systronics micro pH meter model 361 and Deluxe conductivity meter model 601. Total Hardness (TH), Ca2+, Mg2+, Cl−, 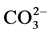 and
and 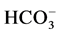 were determined by titration. Na+ and K+ were measured by Flame photometry,
were determined by titration. Na+ and K+ were measured by Flame photometry, 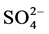 by Lovibond spectrophotometer.
by Lovibond spectrophotometer.
![]()
Figure 1. Map of the study area with water sample locations.
4. Results and Discussion
4.1. Groundwater Chemistry
A summary of the physico-chemical analysis of the groundwater samples is presented in Table 1. The minimum and maximum values of pH and electrical conductivity (EC) of groundwaters are ranging from 7.9 to 8.0 and 230 to 1190 μmhos/cm, respectively. Total dissolved solids (TDS) in the study area vary between 147 to 762 mg/l. The groundwater in the study area is falling under fresh (TDS < 1000 mg/l) types of water (Freeze and Cherry, 1979). The total hardness (as CaCO3) varies from 71 to 446 mg/l. In the study area, the Na and K concentrations in groundwater range from 8 to 138 and 2 to 168 mg/l, respectively. The concentrations of Ca and Mg range from 7 to 72 mg/l and 7 to 64 mg/l, respectively. The Si content varies from 3 to 12 ppm. The concentrations of CO3 and HCO3 ions found in the groundwater samples of study area are ranged from 2 to 72 and 10 to
![]()
Table 1. Minimum, maximum and average values of different constituents of water samples.
209 mg/l respectively. The concentration of Cl ranges from 39 to 306 mg/l. The alkalinity varies from 44 to 230 ppm and the SO4 varies from 10 to 57 mg/l. The abundance of the major ions in groundwater is in following order:
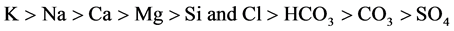
4.2. Hydrochemical Facies for Groundwater
The hydrochemical facies of a particular place are influenced by geology of the area and distribution of facies by the hydro-geological controls.
4.3. Chadha’s Diagram
In the present study, the groundwater of the study area has been classified as per Chadha’s diagram [22] and to identify the hydrochemical processes. The diagram is a somewhat modified version of Piper trilinear diagram [23] and the expanded Durov diagram [24] . In the Chadha’s diagram the difference in milliequivalent percentage between alkaline earth’s (calcium plus magnesium) and alkali metals (sodium plus potassium) expressed as percentage reacting values is plotted on the X-axis and the difference in milliequivalent percentage between weak acidic anions (carbonate plus bicarbonate) and strong acidic anions (chloride plus sulphate) is plotted on the Y-axis (Figure 2). The milliequivalent percentage differences between alkaline earth and alkali metals and between weak acidic anions and strong acidic anions would plot in one of the four possible sub-fields of the diagram. The square or rectangular field describes the overall character of the water. The diagram can be used to study various hydro chemical processes, such as base cation exchange, cement pollution, mixing of natural waters, sulphate reduction, saline water and other related hydro chemical problems [22] . The chemical analyses data of all the samples collected from the study area have been plotted on Chadha’s diagram (Figure 2). It is evident from the results, that 30 samples fall in two groups i.e. Group 6 (53% samples) (Ca-Mg-Cl); and Group 7 (47% samples) (Na-Cl).
4.4. Suitability of Groundwater for Agriculture Purpose
The mean, maximum, minimum and standard deviation of different parameters have been described in Table 1. Six indices [Sodium adsorption ratio (SAR), Adjusted Sodium adsorption ratio (Adj. SAR), Permeability index (PI), Sodium percentage (Na %), Magnesium hazard, and Residual sodium carbonate (RSC)] were used to determine the suitability of groundwater for irrigation activities.
4.5. Sodium Adsorption Ratio (SAR)
Classification of irrigation water with respect to SAR is primarily based on the effect of exchangeable sodium on the physical condition of the soil. However, Sodium-sensitive plants may suffer injury as a result of sodium accumulation in plant tissues when exchangeable sodium values are lower than those effective in causing deterioration of the physical condition of the soil. The sodium Adsorption ratio (SAR), which was calculated for the water samples based on the formula provided by the US salinity Laboratory Staff (1954) [25] . The SAR is defined as the square root of the ratio of the Sodium (Na) to Calcium + Magnesium (Ca + Mg), i.e.
![]()
Figure 2. Chadha’s diagram (modified piper diagram).
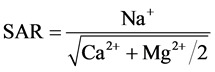 (All ions in epm)
(All ions in epm)
The SAR values of the ground water samples in the study area varied from 0.33 to 4.93 with an average value of 2.27. The SAR values of the water samples of the study area are less than 10, and are classified as excellent for irrigation.
4.6. Integrated Effect of EC and SAR
Richard’s classification of sodium adsorption ratio proposes that the ground waters of the study area are excellent for irrigation. SAR is plotted against EC, which is designated as after U.S. Salinity Laboratory 1954 and is illustrated in Figure 3. This diagram has concluded that the 16 samples (53.33%) falls into C2-S1 category, which indicates medium salinity with low sodium; 13 samples (43.33%) falls in C3-S1, indicates high salinity and low sodium; and one sample in C1-S1 showing low sodium and low salinity. All these waters are being used for irrigation as they posses good soil drainage. Further, these waters are rated as satisfactory for agricultural purposes.
4.7. Adjusted Sodium Adsorption Ratio (Adj. SAR)
The presence of or introduction of bicarbonate and carbonate ions in the irrigation water increases the permeability hazard as quantified by SAR. Irrigation of calcium-rich or magnesium rich soil with water containing
![]()
Figure 3. The quality of water samples in relation to salinity and sodium hazard (after U.S. Salinity Laboratory 1954).
carbonate or bicarbonate ions will form insoluble calcium and magnesium carbonate (limestone, dolomite), thereby reducing the concentration of calcium and magnesium applied to the SAR calculation. This consideration in the calculation of SAR results in the adjusted SAR (Adj. SAR) being greater than the SAR, thereby providing a truer index of the sodicity of the water and the risk of dispersion. Most Adj. SAR values of irrigation waters are about 10 to 15 percent greater than the unadjusted SAR. Additionally, irrigation water with a low salt concentration and a high SAR will contribute to reduced permeability of dispersive soils eventually. This parameter is basically used for assessment of alkalinity hazard in irrigation water and it is ranging from 0.3 to 4.59 (Table 1). The result showed that the concern due to sodium hazard of the water became more emphatic because in all water samples Adj. SAR is higher than SAR. This can be calculated with the following formula:
Adjusted sodium adsorption ratio (adj.SAR) = SAR [1 + (8.4 − pHc)] [26]

where
pK2 Negative logarithm of the second disassociation constant for carbonic acid
pKc Solubility constant for calcite
p Negative logarithm of ion concentration
All ionic concentration is in meql-1
The Adj. SAR values are ranging from 0.44 to 8.10 with a mean value of 3.75. Based on Ayers and Tanji [27] classification all samples have Adj.SAR values are showing <3 and are safe for irrigation.
4.8. Permeability Index (PI)
The soil permeability is affected by long term use of irrigation water. It is influenced by sodium, calcium, magnesium and bicarbonate contents of soil. Doneen [28] has evolved a criterion for assessing the suitability of water for irrigation based on Permeability Index (PI).

The result of study area is ranges from 27.68 to 92.33 with a mean value of 66.25. The majority of the samples fall under class-I under sampling programs of Doneen’s classification, which indicates that groundwater is good for irrigation.
4.9. Percent Sodium (Na %)
Sodium concentration is an important factor in classifying the water for irrigation purposes because it can react with soil resulting in the clogging of particles, thereby, reducing the permeability of water and respiratory gases [11] . Sodium concentration in groundwater is a very important parameter in determining the irrigation quality. Todd [29] defined soluble sodium percentage (SP) as:
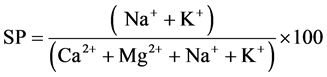
where all the ionic concentrations are in meq.
The percent sodium values of the study area are shown in Table 1 and vary from between 10.29 and 68.95. The classification of groundwater was grouped based on percent Sodium as Excellent (<20%), Good (20% - 40%), Permissible (40% - 60%), Doubtful (60% - 80%) and Unsuitable (>80%) [30] . Out of selected wells, based on percent Sodium, 17% of the wells have excellent irrigation water quality, 23% of the wells have good irrigation water quality, 30% of the wells have permissible irrigation water quality, 30% of the wells have doubtful irrigation water quality.
4.10. Wilcox Diagram
Wilcox [31] used percentage sodium and electrical conductance in evaluating the suitability of groundwater for irrigation. In the Wilcox diagram percent sodium is plotted against conductivity (Figure 4). The diagram consists of five distinct areas such as excellent to good, good to permissible, permissible to doubtful, doubtful to
![]()
Figure 4. The quality water in relation to electrical conductivity and percent sodium (Wilcox diagram).
unsuitable and finally unsuitable. The results (Table 1) show that, 57% of the water samples clustered in the zone of excellent to good; while 27% is plotted at the boundary of good to permissible and the remaining samples in permissible to doubtful category. Therefore, the water samples are good for irrigation.
4.11. Residual Sodium Carbonate (RSC)
It refers to the residual alkalinity and is calculated for irrigation water by the following formula;
![]()
where all ionic concentrations are expressed in epm.
According to the U.S. salinity Laboratory [25] , an RSC value less than 1.25 meq/l is safe for irrigation; a value between 1.25 and 2.5 meq/l is marginal quality and a value greater than 2.5 meq/l is unsuitable for irrigation [32] . The RSC values of groundwater samples of the study area ranges from −5.30 to 0.92 mg/l. Therefore, the groundwaters of the study area are safe for irrigation.
4.12. Magnesium Hazard
Generally, Ca and Mg maintain a state of equilibrium in most waters. More Mg in waters adversely affects the crop yield. As the rocks of the study area comprise dolomites, most waters possess more Mg than Ca. For this reason in this study magnesium hazard was evaluate by two following methods including “Magnesium Ratio” and “Ca to Mg Molar Ratio”. From the Table 1, it is seen that the magnesium ratio {Mg Ratio = [Mg/(Ca + Mg)] × 100} has varied from 19.37 to 87.92 with an average value of 66.25. In this study nearly majority of the water samples has Mg ratio more than 50%, would adversely affect the crop yield as turn the soils more alkaline [33] .
4.13. Kelley’s Ratio (KR)
It is the ratio of sodium ion to calcium and magnesium ion in epm [34] and expressed as; The Kelley’s Ratio (KR) have been computed with the following equation for all groundwater samples of the study area and presented in Table 1.
![]()
where all ionic concentrations are expressed in epm.
In the study area KR ranges from 0.10 to 2.19 with a mean value of 0.95 indicating that water is suitable for irrigation purpose as the average value is less than 1.
4.14. Non-Carbonate Hardness (NCH)
Hardness of water relates to the reaction with soap, since Ca and Mg ions precipitate soap. Hardness is expressed as mg/l of CaCO3. If the hardness as CaCO3 exceeds the difference between the alkalinity as CaCO3 and hardness as CaCO3, it is termed as Non Carbonate Hardness. NCH is also called permanent hardness. From the Table 1 it can be delineated that the NCH values ranged from −46.03 to 264.81 with an average of 66.72.
4.15. Indices of Base Exchange (IBE)
Control of the dissolution of undesirable constituents in waters is impossible during the subsurface run off, but it is essential to know the changes undergone by the waters during its movement [35] . The ion exchange between the groundwater and its host environment during residence or travel are well understood by studying the chloro-alkaline indices. To know the direction of exchange during the path of groundwater through the aquifer, Schoeller [36] [37] , suggested two chloro alkaline indices CaI1 and CaI2 to indicate the exchange of ions between groundwater and its host environment. This is positive when there is an exchange of Na and K from the water with Mg and Ca of the rocks, and is negative when there is an exchange of Mg and Ca of the waters with Na and K of the rocks.
From Table 1 it can be put forth that the CaI1 values range from −0.40 to 0.82 and CaI2 values vary from −0.36 to 1.61. From these values it can be interpreted that some of the samples in the study area fall into negative zones and some fall into positive zones.
4.16. Mechanisms Controlling Groundwater Chemistry
Gibbs diagram [38] , representing the ratios of Na+/(Na+ + Ca2+) and Cl−/(Cl− + HCO3) as a function of TDS, are widely employed to assess the functional sources of dissolved chemical constituents, such as precipitation dominance, rock dominance and evaporation dominance. The chemical data of water samples of the area are plotted in Gibbs diagrams (Figure 5). In the present study all water samples are falling in rock dominance zone. Gibbs diagram reveals that the rock dominance zone indicating the dissolution of silicate bearing rocks with groundwater.
5. Conclusions
The overall quality of waters in the study area rules out any pollution from extraneous sources. As such, the waters are very good for domestic and irrigation uses. The enrichment of constituent elements viz., sodium, potassium, calcium, magnesium and the anions in waters is mainly due to minerals like feldspars, mica, and hornblende.
![]()
Figure 5. Mechanism controlling the chemistry of groundwater (after Gibbs 1970).
These minerals are responsible for the release of above-mentioned elements predominantly in large amounts. These cations are solubilised and removed by leaching, leaving a residue deprived of its easily soluble bases. The primary source for sodium in ground water is due to the weathering of plagioclase feldspar [39] .
The exchange of calcium and magnesium in water by sodium bound in clay and/or cation exchange or HCO3 enrichment possibly from silicate weathering [40] , 1997. Silicate weathering is one of the key geochemical processes controlling the major ions chemistry of the groundwater, especially in hard rock aquifers [41] - [43] . Furthermore, weathering of soda Feldspar (Albite) and Potash Feldspars (Orthoclase and Microcline) may contribute Na+ and K+ ions to groundwater. Feldspars are more susceptible for weathering and alteration than Quartz in Silicate rocks.
Permeability index (PI) occurs when normal infiltration rate of soil is appreciably reduced and hinders moisture supply to crops which is responsible for two most water quality factors as salinity of water and its sodium content relative to calcium and magnesium. Highly saline water increases the infiltration rate. Based on the USSL diagram, the waters are classified with reference to SAR. In general, most of the samples are showing medium and high salinity hazard with low sodium hazard. According to Wilcox diagram, majority of samples are falling in the fields of excellent to good and good to permissible categories. The chloroalkalinity index is used to evaluate the extent of base exchange during rock-water interaction.
The groundwater samples have also been evaluated for their irrigation quality. The parameters viz., Sodium adsorption ratio (SAR), Adjusted Sodium adsorption ratio (Adj. SAR), Permeability index (PI), Sodium percentage (Na %), Magnesium hazard, and Residual sodium carbonate (RSC) suggest that groundwater of the study area is suitable for irrigation purpose. The Chada’s diagram has revealed that the majority of the samples are falling in Na-Cl type and Ca-Mg-Cl types.
NOTES
![]()
*Corresponding author.