Polariton Evaporation: The Blackbody Radiation Nature of the Low-Frequency Radiation Emitted by Radiative Polaritons to the Surrounding Space ()
1. Introduction
Radiative polaritons (RPs) were discovered in the late sixties [1] -[3] and recently gained attention due to their ability to explain optical and thermal properties of thin oxide films or crystals [4] -[11] . Radiative polaritons form upon the absorption of photons from infrared (IR) radiation by phonons in thin oxide films or crystals. The coupling occurs when photons and phonons oscillate at the same frequency. Unlike surface phonon-polaritons [12] , whose frequency is a real number, RPs are characterized by a complex angular frequency
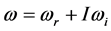
, where
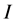
is the imaginary unit and the subscripts
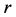
and
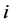
refer to the real and imaginary parts, respectively [3] . The real part,
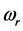
, is the resonant frequency, and is larger than the imaginary part,
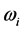
[13] . Additionally, through IR absorption spectra,
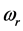
provides the central frequency of the absorption peak, while
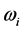
supplies the spread of the absorption peak around
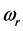
[2] [3] [13] , or half of the peak width. It was recently found that the frequency
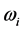
corresponds to the frequency of the experimentally observed low-frequency radiation emitted to the space surrounding the RP’s formation site [13] . Such radiation lasts as long as the exciting IR radiation illuminates the targeted thin oxide film or crystal. The existence of such low-frequency radiation, however, is so far only viewed as the consequence of the presence of
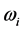
in one of the exponential terms in the expression for the polarization
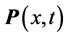
:
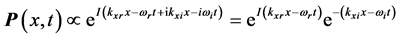
, (1) where
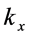
is a component of the complex wave-vector
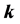
parallel to the thin oxide film or crystal surface [2] [12] . The term
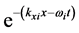
in Equation (1) generates a wave propagating away from the site of the thin oxide film or crystal that generated the RP without decay. This description gives merely a mathematical explanation without providing a physical mechanism of the origin of the low-frequency radiation with frequency
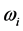
. Furthermore, no explanation is provided for the wide span of the
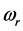
values, on one hand, and the confinement of the
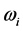
values in a small frequency range in the microwave to far-IR region for a large variety of thin oxide films in a broad thickness range. The goal of this work is to unveil the explanation (1) of the physical mechanism underpinning the formation of the low-frequency radiation with frequency
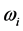
, and (2) for the wide span of the
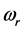
values, on one hand, and the confinement of the
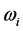
values. The importance of this effort is in its ability to elucidate whether a new source of radiation accompanies the formation of RPs, or if a connection can be found with already known phenomena.
2. Experimental Data and Simulation Method
The experimental data consist of the IR spectroscopic information on thin oxide films reported by previous research. From IR absorption spectra, the frequency
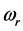
can be derived from the centroid of the absorption peak, while the frequency
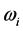
is derived from half of the width of the absorption peak [2] [3] [13] , as illustrated in
Figure 1.
Table 1 summarizes the findings obtained for the 0TH type RP [2] [3] of various oxides, such as TiO
2, La
2O
3, Al
2O
3, and Lu
2O
3 [13] -[17] . It is found that, for the 0TH type RP, the
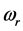
values span in
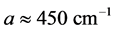
wide range, while the corresponding
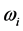
values span in a range
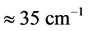
wide. The range of the
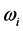
values is
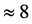
% that of the
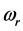
values. In addition, the
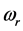
values are very characteristic of a peculiar crystal structure

Figure 1. Experimental absorptance spectra at a 60˚ incidence angle for a 250 nm thick Al2O3 film on Si(100) measured in reflection mode [13] . The 0TH type RP is labeled with the frequency  at the centroid of the absorption peak, and the frequency
at the centroid of the absorption peak, and the frequency  indicating the width of the absorption peak [13] .
indicating the width of the absorption peak [13] .
[1] [3] [18] , which can be determined using IR spectroscopy. The
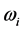
values, instead, are confined in the microwave to far-IR region of the electromagnetic frequency spectrum [13] , as shown in
Table 1, for a large variety of oxides and in a broad thickness range. In this research, the spectroscopic data collected in
Table 1 are used and elaborated according to the hypothesis formulated in the following section (Section 3).
3. Hypothesis
For a large variety of thin oxide films in a broad thickness range, the understanding of the wide span of the
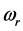
values, on one hand, and of the confinement, on the other, of the
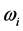
values requires a new approach beyond the analysis of the polarization
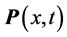
, as done in previous research [2] [13] . The alternative path chosen here is of statistical mechanics nature. It involves photons and phonons, which are mass-less bosons obeying the general Bose-Einstein statistics:
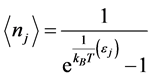
. (2)
Here,  is the occupation number of bosons with energy
is the occupation number of bosons with energy ,
,  the Boltzmann constant, and
the Boltzmann constant, and  the temperature. Stemming from the coupling between photons and the phonons, RPs must be bosons. The formation of a RP can be described as the result of an annihilation operator
the temperature. Stemming from the coupling between photons and the phonons, RPs must be bosons. The formation of a RP can be described as the result of an annihilation operator  applied to both the Hamiltonians of the photon and phonon, and contemporarily a creation operator
applied to both the Hamiltonians of the photon and phonon, and contemporarily a creation operator  applied to the Hamiltonian of the RP. Alternatively, the effort made by a thin oxide film or crystal to couple photons and phonons and generate a RP can be expressed in terms of the chemical potential
applied to the Hamiltonian of the RP. Alternatively, the effort made by a thin oxide film or crystal to couple photons and phonons and generate a RP can be expressed in terms of the chemical potential . In this context,
. In this context,  is thus defined as the free energy needed to rise or lower the number or moles of RPs in a thin oxide film or crystal. Assuming that
is thus defined as the free energy needed to rise or lower the number or moles of RPs in a thin oxide film or crystal. Assuming that  is the energy of a RP, and considering
is the energy of a RP, and considering  as the number of moles of RPs, the Bose-Einstein statistics for RPs is:
as the number of moles of RPs, the Bose-Einstein statistics for RPs is:
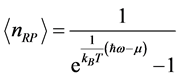
, (3) where
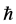
is the reduced Planck’s constant (
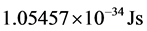
). It follows that the temperature
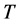
associated to the RP can be derived from Equation (3) as:
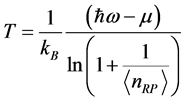
, (4) where
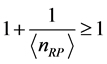
,
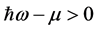
, and
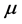
is real and positive. Furthermore, since
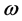
is complex, the temperature
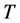
must be complex too. The real part:
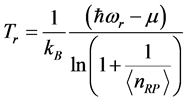
, (5) is related to the frequency of the radiation absorbed at
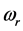
and to the chemical potential
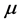
, whereas the imaginary part:
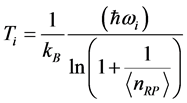
, (6) is associated with the low-frequency radiation emitted at frequency
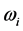
. For both the real and the imaginary parts, Equation (3) links
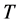
to
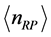
, the number of moles of RPs, and to the magnitude of the characteristic RPs frequencies,
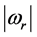
and
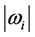
respectively. The real and an imaginary temperatures
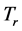
and
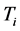
coexist in a system of thin oxide films or crystals, and their environment, while accompanying RP’s formation. The investigation will now proceed with discussing the physical meaning of
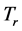
and
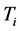
, and their relationship with the origin and properties of the low-frequency radiation emitted at frequency
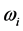
.
4. Results and Discussion
Equation (5) provides an expression for the real temperature,
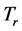
, whose nature needs to be disclosed alongside the value of the chemical potential
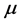
. Two possibilities can be considered. One is that the value of the chemical potential
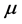
is the same for the RPs in all types of a thin oxide film or crystal that can support them. In this case, according to Equation (5),
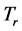
depends on the oxide chemistry, thus on
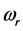
. This picture rules out the influence on the results due to the laboratory temperature, which determines the number of moles for RPs
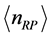
at a given frequency [19] , and will therefore be neglected. The alternative possibility is that the value of the chemical potential
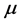
varies for different types of thin oxide films or crystals. In this case,
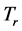
can be constant, and taken as the laboratory temperature at which the RPs were generated. The data in
Table 1 were collected at
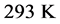
, or 20˚C, which is thus assumed as the laboratory’s temperature. In this case, the chemical potential
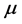
values vary depending upon the thin oxide films considered, and their values in
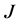
are reported in
Table 1 assuming
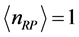
. Low
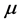
values characterize a crystal structure and chemistry which require a small amount of energy to promote RPs formation. The span in the
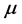
values explains the large span of the
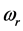
values measured for thin oxide films. The trend of the chemical potential
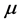
versus
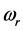
:
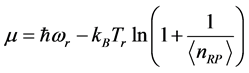
, (7) is illustrated in
Figure 2. It can be observed that larger chemical potential
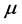
values are associated to larger absorption frequencies
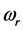
.The trends are similar for
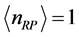
and
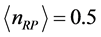
with
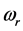
values ranging between
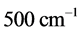
and
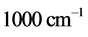
, as observed in experimental spectra. Equation (6) gives the expression for the imaginary temperature
. This expression is similar to
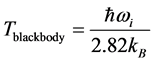
[20] , the temperature of blackbody radiation with maximum spectral radiance
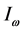
at
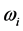
. The factor
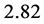
stems from the maximization of
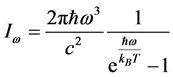
with respect to
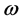
[20] , where
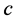
is the speed of light in vacuum. The similarity between Equation (6) and
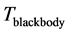
suggests two possible conse- quences. One is that that the radiation emitted to the space surrounding the RP’s formation site at frequency
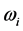
is blackbody radiation. Thus, the association between
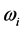
and the imaginary temperature
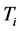
in Equation (6) is named polariton evaporation, which can be viewed as the emission of energy, or as some kind of dissipation phenomenon, related to the frequency
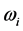
. Stated differently, the dissipated RP’s energy
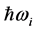
plus the energy from the IR radiation absorbed around frequency
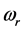
and transformed into heat [21] , give the energy of the IR
photons absorbed by the thin oxide film or crystal. The other consequence is that, for
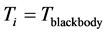
,
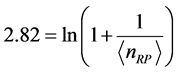
. (8) Equation (8) has a solution for
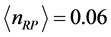
. Since here the magnitude of
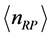
is a fractional value, this quantity should not be related to number of particles. Rather, defining
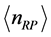
as the mole number of RPs seems more appropriate. With this assumption, the trend of
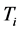
versus
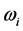
between
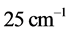
and
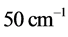
is illustrated in
Figure 3 for
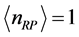
and
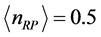
. The
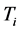
temperature values increase linearly and with positive slope with respect to
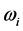
. The values of
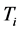
are around or below 73 K, or −200˚C, and vary by only 40˚C to 20˚C for
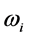
between
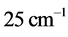
and
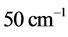
for
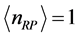
and
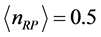
, respectively. Further more, for
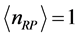
the slope is
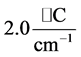
, while for
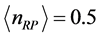
it is
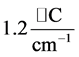
. The dependence of the
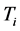
values to the number of moles is related to the dependence of the
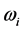
values to film thickness, or the amount of material in the thin oxide film, as discussed in previous research [13] . The proximity of the
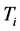
values to
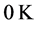
, or
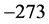
˚C, combined with the effects of the number of moles of the thin oxide film, confines the
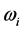
values in a small range in the microwave to far-IR region of the electromagnetic frequency spectrum [13] , as shown in
Table 1. This observation also signifies that detecting T
i is not trivial: it would require a thermometer operating near
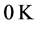
, and sensitive to very small variations of temperature.
5. Comparison with Other “Evaporation” Phenomena
Evaporation phenomena and imaginary temperature in the available literature are now discussed. Evaporation phenomena in particular are not isolated, and one of the most popular is black hole evaporation [22] [23] . The emission of the so-called Hawking radiation was hypothesized after the discovery that black holes have entropy, and thus temperature. Because of the temperature, black holes must radiate blackbody radiation. Unlike the polariton evaporation case, the temperature of black holes was discovered before the radiation. The temperature of the black hole is a real, not an imaginary quantity [22] [23] . The evaporation phenomenon for black holes is of quantum-mechanical nature, and involves the “evaporation” of mass [23] in the form of particle production via
tunneling [24] . Polariton evaporation, on the other hand, is the consequence of a complex frequency, which results in a complex temperature. Thus, even if polariton evaporation is not accompanied by real mass loss, the complex frequencies and temperature are related to the existence of dissipation events [13] [25] . The energy balance is such that the sum of the energy
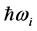
dissipated by the RPs plus the energy from the IR radiation absorbed around frequency
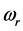
and transformed into heat [21] , are equal the energy of the IR photons irradiating the thin oxide film or crystal. Similarly to the case of the imaginary temperature
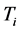
of polariton evaporation, which is relatively low
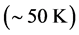
, the temperature of the black hole is predicted to be low (
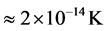
for a supermassive black hole) [26] . Investigations are under way to detect the existence of the Hawking radiation, whose signature is contained in gamma-ray bursts [23] . On the other hand, the radiation related to polariton evaporation was recently detected [13] . Finally, similarly to the temperature of the black holes which depends on
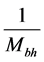
, where
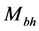
is the mass of the black hole [23] , the
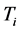
of RPs depends onthe amount of RPs in the form of their number of moles
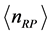
, as shown in
Figure 3. Even with all the differences reported, polariton and black hole evaporation are both related to blackbody radiation, showing a deep unity in microscopic and macroscopic physical and natural phenomena. The concept of imaginary temperature is not as popular as that of “evaporation”. The literature is short of examples of imaginary temperatures deriving from mathematically complex quantities. On the other hand, the expression of imaginary temperature is used most notably in the biological field, where it is defined as the “statistical value for a thermostat made of particles with real mass” [27] . Mathematically imaginary quantities, however, are normally related to dissipation [13] [25] .
6. Summary and Significance
Because of their bosonic nature, radiative polaritons have a temperature associated with the low frequency radiation they emit to the space surrounding their formation site. The radiation is due to blackbody radiation associated with a temperature which stems from the imaginary part of the complex frequency of radiative polaritons and is related to their amount. The relationship with blackbody radiation aids in explaining the confinement of the
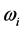
values in a narrow frequency interval in the microwave to far-infrared region. The
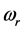
values span in a wide frequency range because they are related to the oxide film and crystal properties, as illustrated here specifically for the 0TH type radiative polariton in
Table 1. Finally, polariton evaporation resembles black hole evaporation, giving insight on the profound unity among physical phenomena in the nanoand the macro-scale.
Acknowledgements
This work was supported by the Thomas F. Jeffress and Kate Miller Jeffress Memorial Trust (grant # J-1053), the James Madison University (JMU) Center for Materials Science, the JMU Department of Physics and Astronomy, and the NSF-REU and Department of Defense ASSURE program (grant #0851367). The authors thank Profs. K. Fukumura, A. Constantin, C. S. Whisnant, and J.C. Zimmerman (JMU) for fruitful discussions.
NOTES
*Corresponding author.