Determination and Quantification of Vanadium(V) in Environmental Samples Using Chemically Modified Chitosan Sorbent ()
1. Introduction
Vanadium compounds are released in large quantities from burning fossil fuels and from various industrial processes [1] . These are precipitated on the soil drained by rain and groundwater and directly adsorbed by plants, animals and humans. Vanadium content in food is directly dependent upon the concentrations present in the soil. Whereas, the concentration of vanadium in water is largely dependent on geographical location and ranges from 0.2 to >100 µg∙L−1 in fresh water and drinking water, from 0.2 to 29 µg∙L−1 in seawater and the typical values lies between 1 and 6 µg∙L−1 [2] -[4] . The toxicity of vanadium is dependent on its oxidation state, with vana- dium(V) being more toxic than vanadium (IV) and its poisoning symptoms are sometimes fatal [5] . Industrial discharges in application of vanadium such as dyeing, ceramics, ink and catalyst manufacturers contribute to its presence in water supply [6] . Vanadium in soils at concentrations of 10 mg∙kg−1 or more is toxic for terrestrial plants [7] . The threshold limit values (TLV) reported are 0.5 mg/cubic meter of air and 0.1 mg/cubic meter of fume [8] . Therefore, the determination of vanadium in environmental samples is necessary in terms of environ- mental protection. Several analytical techniques have been reported for the determination of vanadium, which include spectrophotometry [9] - [15] , neutron activation analysis (NAA) [16] [17] , inductively coupled plasma mass spectrometry (ICP-MS) [18] , inductively coupled plasma optical emission spectrometry (ICP-OES) [1] [19] , atomic absorption spectrometry [20] [21] , in various environmental samples. However, most of these tech- niques suffer from several setbacks such as, multiple spectral interferences in spectrophotometric methods, few techniques like ICP-MS and ICP-OES are expensive, NAA method is time-consuming and requires sophisti- cated instrumentation and atomic absorption spectrometric (AAS) determinations are either insensitive (flame) or non selective due to pronounced matrix interference (graphite furnace). The direct analysis of vanadium in complex matrices is difficult as they occur at very low concentrations. In order to achieve accurate and sensitive results, a suitable preconcentration step is required to address these problems in the analysis of real samples.
The use of chitosan to treat low concentration of heavy metals from environmental samples has grown re- cently because of its high adsorptive capacity when compared to other adsorbents [22] .
In this work, a rapid sensitive and selected method is described for preconcentrative determination of vana- dium(V) using the synthesized 3-Hydroxybenzaldehyde-4 Amino antipyrine, which was chemically immobi- lized on chitosan.
2. Materials and Methods
2.1. Chemicals and Reagents
All the reagents used in this study were of analytical grade and standard solutions of vanadium(V) were pre- pared by dissolving appropriate amounts of ammonium metavanadate (99.99%, Merck) in double distilled water. The 0.1 mol∙L−1 acetic acid/acetate buffer solution was prepared by adjusting the pH to 3.5. Commercially available chitosan was used for the present study.
2.2. Synthesis of 3-Hydroxybenzaldehyde-4-Amino Antipyrine
The 3-Hydroxybenzaldehyde-4-Amino antipyrine was synthesized by refluxing equimolar amount of 3-Hydro- xybenzaldehyde (99%, Merck) and 4 Amino antipyrine (99%, Merck) in methanol for 3 hours at 40˚C [23] . The product was recrystallized from each solvent used in the synthesis and dried at room temperature. The product (HBAP) was characterized by infrared spectroscopy using KBr pellet method.
About 1.6268 grams of 3-Hydroxybenzaldehyde-4-Amino antipyrine synthesized product was dissolved in Dimethylformamide in 100 mL volumetric flask, from this a known volume of 0.05 mol∙L−1 HBAP was used for the determination of vanadium(V).
2.3. General Procedure
To the known concentration of vanadium(V), standard solutions were adjusted to pH = 3.5 ± 0.2 by adding 3 mL of acetic acid acetate buffer. An amount of 30 mg of chitosan and 0.5 ml of synthesized 0.05 mol∙L−1 3-Hy- droxybenzaldehyde-4-Amino antipyrine was added to the above solution and stirred for about 30 minutes and the solution was filtered by using vacuum filtration. The filtered solution was eluted with 20 ml of 1 mol∙L−1 HCl solution and then the vanadium is determined by Flame atomic absorption spectrometer (FAAS).
2.4. Apparatus
Measurements were performed with a model 6300 Flame atomic absorption spectrometer (FAAS), Shimadzu (Japan), with flame of N2O-C2H2 and the instrument settings were according to the manufacturer’s recommendations. A LI-120 digital pH meter (Elico, India) was used for the pH measurement and reagents and chemicals were weighed using analytical balance, Shimadzu AUX 320.
UV-visible spectrophotometer model UV-1800 from Shimadzu (Japan), was used for recording the absor- bance spectra for the complexation studies of vanadium(V). IR spectrometer (Thermo-Nicolet FT-IR, Nicolet IR-200, USA) was used for the analysis of functional groups in the synthesized reagent.
2.5. Procedure for Soil Analysis
Soil samples were collected in and around industrial sites of Tirupati. The collected soil samples were air dried at 70˚C in the laboratory oven and then grinded to fine powder and then sieved through 0.25 mm nylon mesh. To one gram of soil sample, 8 mL concentrated HCl and 2 mL concentrated HNO3 were added and kept for overnight [24] . After digestion and filtration, the solution was subjected to separation by following the general procedure described above and subjected to FAAS for determination of vanadium(V).
2.6. Application to Real Samples
The developed procedure was used for the quantification and determination of V(V) ions in the real water and soil samples. The water and soil samples were collected in and around industrial sites of Tirupati. To the 20 mL of water sample (ground water/tap water) and acid digested soil sample followed by the general procedure de- scribed above and finally subjected to FAAS for the determination of vanadium(V).
2.7. Interference Study
The interference of foreign ions like Cl−, F−, 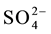 ,
, 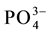 , Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ was studied by equilibrating the fixed amount of vanadium(V) along with the reagent sorbent so- lution at pH = 3.5 and later the determinations was made by FAAS.
, Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ was studied by equilibrating the fixed amount of vanadium(V) along with the reagent sorbent so- lution at pH = 3.5 and later the determinations was made by FAAS.
3. Results and Discussion
3.1. Characterization of Developed Sorbent
The synthesized reagent 3-Hydroxybenzaldehyde-4-Amino antipyrine (HBAP) immobilized on the surface of chitosan was found to complex with vanadium(V) ions at pH = 3.5. This metal complexation with HBAP is due to its phenolic rings leaving the hydrophilic moieties (oxygen and azo-nitrogens) that are available for metal complexation through the formation of one or two coordination spheres leading to the respective stable structure. This seems to suggest that terdentate ligand (HBAP) is assumed to form 1:2 metal-HBAP complex in the de- veloped preconcentration step as shown in Scheme 1.
The FTIR spectra of 3-Hydroxybenzaldehyde-4-Amino antipyrine (HBAP) were recorded and are shown in Figure 1. Here the vibration bands at 3424.30 cm−1, 1630.41 cm−1, 2919.44 cm−1 and 1517.17 cm−1 are due to the presence of hydroxyl group, Carbonyl (C=O), C-H and presence C-N stretching vibrations respectively.
The reaction of HBAP solution with vanadium(V) cation forms a pink colour with a maximum absorbance at 525 nm as shown in Figure 2. The blank HBAP reagent has an absorption maximum at 383 nm, whereas the HBAP-vanadium(V) complex reaches a maximum after 10 min and this is stable for at least 2 h.
3.2. Experimental Optimum Conditions
The uptake efficiency of 5 µg∙L−1 vanadium(V) in 20 mL of HBAP solution was studied as a function of pH. As seen from the Figure 3, the decrease in percentage enrichment at pH less than 3.0 and above 4.0 is due to the insufficient formation of the vanadium(V)-HBAP complex. Thus, the optimum pH = 3.5 was selected through- out the experiment by adjusting the pH using acetic acid acetate buffer.
To know the optimum reagent concentration, 0.05 mol∙L−1 HBAP reagent solution prepared using dimethyl- farmamide ranging 0.3 mL to 1 mL was used to know the optimum reagent concentration. Absorbance increases with increase of volume of HBAP reagent up to 0.4 mL and thereafter it remains constant. Hence further studies were carried out using 0.5 mL of HBAP reagent. The weight of chitosan sorbent was tested ranging from 10 mg to 50 mg and found that 30 mg of sorbent was sufficient for the effective preconcentration of vanadium(V).
![]() M = vanadium(V).
M = vanadium(V).
Scheme 1.Chemical structure of metal-HBAP complex in 1:2 ratio.
![]()
Figure 1. FTIR spectrum of the synthesized 3-Hydroxybenzaldehyde-4 Amino antipyrine (HBAP) reagent.
![]()
Figure 2. Absorption spectra of HBAP and its vanadium(V) complex.
![]()
Figure 3. % enrichment of vanadium(V) as a function of pH in fixed amount of 5 µg∙L−1 of vanadium(V).
Various reagents such as 1 mol∙L−1 HCl, 1 mol∙L−1 HNO3 and 1 mol∙L−1 H2SO4 were tested for eluting the com- plexed vanadium(V) for its determination by FAAS. It was found that 20 mL of 1 mol∙L−1 HCl could effectively elute vanadium(V) from the HBAP chitosan sorbent. The equilibration time required for the better enrichment of vanadium(V) was found to be 30 min. The optimization conditions of preconcentration of vanadium(V) were shown in Table 1.
3.3. FAAS Analysis and Its Analytical Performance
It was observed from FAAS analysis, that the response of the eluted vanadium(V) from equilibrated HBAP chi- tosan sorbent was linear in the concentration range of 0.5 µg∙L−1 - 7 µg∙L−1 vanadium(V) as shown in Figure 4 with a detection limit of 0.1 µg∙L−1. These values are 100 times lower than by direct flame atomic absorption spectrometry (FAAS) method after preconcentration with HBAP chitosan sorbent. The precision of the devel- oped procedure is considered good with a relative standard deviation of 2.84%. The unknown vanadium within this range can be quantified from the calibration graph obtained.
3.4. Interference Studies and Application to Real Samples
In order to study the selectivity, the HBAP chitosan sorbent was equilibrated with a fixed amount of 0.01
![]()
Table 1. Optimization of experimental parameters during preconcentration and elution of 5 µg∙L−1 of vanadium(V).
![]()
Figure 4. Calibration curve obtained from measurement of absorbance of eluted vanadium(V) by flame atomic absorption spectrophotometry.
mol∙L−1 vanadium(V) and with increasing amounts of ions namely Cl−, F−, 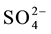 ,
, 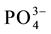 , Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ at pH = 3.5. The absorbance of the eluted sample from the HBAP chitosan sorbent, which was equilibrated with solution containing interfering ions and vanadium(V) was com- pared with the absorbance of eluted sample from the HBAP chitosan sorbent, which was equilibrated in the ab- sence of interfering ion with the solution having the same amount of vanadium under similar experimental con- ditions using FAAS. The tolerance limit of foreign ions in the determination of 0.01 mol∙L−1 vanadium(V) was summarized in Table 2.
, Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ at pH = 3.5. The absorbance of the eluted sample from the HBAP chitosan sorbent, which was equilibrated with solution containing interfering ions and vanadium(V) was com- pared with the absorbance of eluted sample from the HBAP chitosan sorbent, which was equilibrated in the ab- sence of interfering ion with the solution having the same amount of vanadium under similar experimental con- ditions using FAAS. The tolerance limit of foreign ions in the determination of 0.01 mol∙L−1 vanadium(V) was summarized in Table 2.
The applicability of the developed sorbent was examined by spiking 2 µg∙L−1 of vanadium(V) in different ground waters and 2 µg∙g−1 of vanadium(V) in soil samples and the preconcentrated vanadium(V) was deter- mined by FAAS. The recovery obtained in the range 97.5 to 102.5 as shown in Table 3(a) and Table 3(b), seems to suggest that the developed sorbent is feasible for real sample analysis.
4. Conclusion
HBAP immobilized chitosan sorbent was prepared and used as a potential sorbent for preconcentration and se- paration of vanadium(V) prior to their determination. The complexation of vanadium(V) in HBAP chitosan sor- bent was studied as a function of pH and found that the efficiency was 100% at pH = 3.5. The equilibration time required for the better enrichment of vanadium(V) was found to be 30 min and the complexed vanadium can be successfully eluted using 20 mL of 1 mol∙L−1 HCl for FAAS analysis. The calibration plots were linear over the concentration range of 0.5 to 7 µg∙L−1 vanadium(V). From the study of selectivity of the developed sorbent, it was found that vanadium(V) can be preconcentrated from complex aqueous medium in presence of other ions like Cl−, F−, 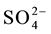 ,
, 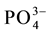 , Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ at pH = 3.5. The preconcentration of vanadium(V) ions from soil and water samples indicated the possibility of using the HBAP chitosan sorbent investigated in the present work for real samples.
, Na+, Ca2+, Zn2+, Fe3+, Fe2+, Cu2+, Cr3+, EDTA, Mn2+, Co2+ and Ni2+ at pH = 3.5. The preconcentration of vanadium(V) ions from soil and water samples indicated the possibility of using the HBAP chitosan sorbent investigated in the present work for real samples.
Acknowledgements
The author G.R.K. Naidu thanks UGC, New Delhi, India for financial assistance of the project under UGC-BSR
![]()
Table 2. Tolerance limit of ions in the determination of 0.01 mol∙L−1 vanadium(V) in aqueous sample at pH = 3.5.
![]() (a)
(a) ![]() (b)
(b)
Table 3. (a) Determination of vanadium(V) in soil samples; (b) Determination of vanadium(V) in water samples.
Faculty Fellowship grant.
NOTES
*Corresponding author.