Thermodynamic dissociation constants pKa of 2,2-bis(hydroxymethyl)-2,2’,2”-nitrilotriethanol have been determined at 12 temperatures from (278.15 to 328.15) K including the body temperature 310.15 K by the electromotive-force measurements (emf) of hydrogen-silver chloride cells without liquid junction of the type: Pt(s), H
2(g), 101.325 kPa|BIS-TRIS (m) + BIS-TRIS·HCl (m)| AgCl(s), Ag(s), where m denotes molality. The pK
a values for the dissociation process of BIS-TRIS·H
++ H
2O = H
3O
+ + BIS-TRIS given as a function of T in Kelvin (K) by the equation pKa = 921.66 (K/T) + 14.0007-1.86197 ln(T/K). At 298.15 and 310.15 K, the values of pK
a for BIS-TRIS were found to be 6.4828 ± 0.0005 and 6.2906 ± 0.0006 respectively. Thus buffer solutions composed of BIS-TRIS and its hydrochloride would be useful as secondary pH buffer standards and for control of acidity in the pH range 6 to 8. At 298.15 K the thermodynamic functions
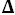
G°,
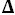
H°,
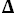
S° and
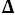
C
p° for the dissociation process of BIS-TRIS·H+ are
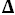
G°=37,005 J·mol
-1,
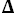
H° = 28,273 J·mol
-1,
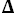
S°= 29.3 J·K
-1·
mol
-1 and
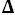
C
p° = 36 J·K
-1·mol
-1. These results are compared with the dissociation of protonated bases structurally related to BIS-TRIS·H
+.