Influence of the Storage Duration at Different Temperatures on the Concentrations of Extractable Inorganic Soil Nutrients ()
Received 18 February 2016; accepted 2 May 2016; published 5 May 2016

1. Introduction
Soil chemistry changes through the time, as biological and chemical processes break down or combine compounds through the time. These processes change once the soil is removed from its natural ecosystem (flora and fauna that penetrate the sampled area) and environment (temperature, moisture, and solar light/radiation cycles). As a result, the chemical composition analysis accuracy can be improved if the soil is analyzed soon after its extraction [1] .
The soil-fresh sample received in the laboratory should be analyzed directly after sampling for determination of nitrate, nitrite and ammonium. The chemical changes in the soil can be slowed during storage and transportation by freezing it. Air-drying can also preserve the soil sample for many months. Lag time between field sampling and analysis must be minimized. Otherwise, storage time will inevitably introduce an additional factor influencing analysis results [2] .
Soil samples are often store before starting the physiochemical properties analysis. This can affect subsequent concentrations of extractable inorganic nutrients, including 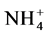 and
and 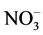 [3] , K+ [4] and
[3] , K+ [4] and 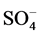 [5] . Moreover, recent study reports that, at 10˚C the values of soluble potassium (K+), calcium (Ca2+), and magnesium (Mg2+) are increased to 10% [6] .
[5] . Moreover, recent study reports that, at 10˚C the values of soluble potassium (K+), calcium (Ca2+), and magnesium (Mg2+) are increased to 10% [6] .
Sample handling and storage are more important aspects of soil analysis than generally recognized. [7] presents data indicating problems associated with drying and storing soil samples and changes that occur under various sample and storage conditions.
In this study, we assessed the influence of short storage periods at different temperatures on the concentrations of extractable soil cations (Ca2+, Mg2+, Na+ and K+) and anions (Cl−,  and
and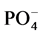 ) in soils of the Nile river terraces at River Nile State, North of Sudan. We tested the hypotheses that i) extractable inorganic nutrients concentrations would be significantly change during different storage periods as well as different temperatures; ii) extractable inorganic nutrients concentrations would be positively correlated with soil formed at different time but derived from similar parent materials; iii) chloride (Cl−) and sulphate (
) in soils of the Nile river terraces at River Nile State, North of Sudan. We tested the hypotheses that i) extractable inorganic nutrients concentrations would be significantly change during different storage periods as well as different temperatures; ii) extractable inorganic nutrients concentrations would be positively correlated with soil formed at different time but derived from similar parent materials; iii) chloride (Cl−) and sulphate (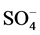 ) concentrations would be most changed during storage periods at different temperatures.
) concentrations would be most changed during storage periods at different temperatures.
2. Materials and Methods
2.1. Site Description
The study area is located in North of Atbara city, River Nile State, Sudan, with an altitude of (336 - 358 meters) above sea level. The study area covered about 8000 ha and is located where the coordinate is at 17.82289 to 17.82389N and 33.99974 to 34.02127E. According to [8] , the study area falls within the arid climatic zone. The average annual rainfall varies from 0 to < 100 mm. Mean maximum temperature of the hottest months (May and June) is 43˚C. Mean minimum temperature of the coldest month (January) is less than 13˚C. The mean annual relative humidity ranges between 15% to 21% (January to February), less than 15% (March to June). The predominant natural vegetation in the study area consists of the following species: Tundub (Capparis decidua), Seyal (Acacia tortilis), Usher, Musket (Prosopis chilensis), Heglig (Balanites aegypiaca) and Seder (Zizyphus spina-christi). The calculated soil temperature regime is hyperthermic and soil moisture regime is arridic. According [9] , the soils of the study area belong within Entisols and Aridisols orders.
2.2. Soil Sampling and Preparation
Nine soil samples were collected from different Nile river terraces at River Nile State, North of Sudan (Table 1). At each site, approximately 5 kg of soil sample was collected from the depth of 0 - 30 cm using an auger and kept in a plastic bag. In the laboratory, soil samples were air-dried and passed through a 2 mm mesh sieve to obtain the fine earth fraction. To estimate the influence of storage period at different temperatures on the concentrations of soluble cations and anions, a 1:5 soil suspension was shaken for 30 minutes, and then extracted
![]()
Table 1. Description of samples sites in the study area and some physical and chemical properties.
with filter paper (Whatman No. 42). Each soil extract then divided into three treatments: i) control (time zero) or immediately analyzed); ii) storage for 10 days and; iii) storage for 30 days. Each treatment was replicated three times: i) storage at 10˚C; ii) storage at ambient laboratory temperature (25˚C) and; iii) storage at 45˚C in incubator.
2.3. Estimation of Soil Properties
The particle size distribution of the subsamples was determined using Particle size analyzer model (Mastersizer 2000, Malvern) and the soil texture class was determined using USDA Textural triangle [10] . Soil pH was measured in 1:5 soil suspensions using a digital pH meter Model (3510, Jenway) and the results were compared according to the classification of [11] . The electrical conductivity (EC dS/m at 25˚C) was determined in 1:5 soil extract using a conductivity meter Model (4510, Jenway) [12] and the results were compared according to the classification of [13] . Percent calcium carbonate (%CaCO3) was estimated by Calcimeter. The samples were treated with 0.1N HCL; the volume of CO2 from pure calcium carbonate and samples were recorded. The percent calcium carbonate was then calculated according to [14] . Extractable cations (Ca2+, Mg2+, Na+ and K+) and anions (Cl−, 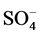 and
and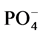 ) were determined in 1:5 soil extraction.
) were determined in 1:5 soil extraction.
2.4. Statistical Analysis
Changes during storage period at 10 and 30 days were determined by repeated measures analysis of variance using storage period and treatment (10˚C, 25˚C and 40˚C) as factors. Values for fresh extracts (control) were compared with those from extracts stored for 10 and 30 days by one-way analysis of variance with Turkey’s significant difference test for mean separation (P < 0.05). All statistical analysis was performed using SPSS software version 16.0 (SPSS Inc, USA).
3. Results and Discussion
3.1. Selected Chemical and Physical Properties
The description of the samples sites in the study area and some of the physical and chemical properties are presented in Table 1 and Table 2 respectively. The soil texture ranged from sandy loam in the first terrace, silt loam in the second terrace (Instructional farm). While the soil texture in the Agricultural College farm was sand. This could be due to the fact that these soils falls in the mining area which are truncated soils. The soils of the study area were moderately calcareous and the %CaCO3 ranged from 2.58% to 7.40%. The soil reaction (Soil pH) varied from alkaline to strongly alkaline with a pH value ranged from 7.46 to 8.80 [11] . According to [13] , the soils of the study area were non-saline and the electrical conductivity (ECe) ranged from 0.22 to 0.57 dS/m. The predominant soluble cations and anions in the study area were (Na+ and Ca++), (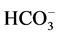 and Cl−) respectively.
and Cl−) respectively.
3.2. Influence of the Storage Period on the Concentrations of Extractable Nutrients
Comparing first and second storage period there were significant difference at P < 0.05 found in all estimated cations (Table 3 and Figure 1) respectively, except for Na+ and K+. The Ca2+ and Mg2+ concentrations were significantly increased from11.22 ± 1.978a to 17.503 ± 4.047b and from 3.790 ± 2.794a - 5.568 ± 3.722b respectively. Similar trend were found when comparing 10 days with 30 days storage period, Ca2+ and Mg2+ concentrations were significantly increased from 17.503 ± 4.047b to 23.505 ± 5.998c in addition to K+ concentration
![]()
Figure 1. Change in soluble soil cations concentrations at different storage periods.
![]()
Table 2. Selected soil chemical properties of soils of the study area.
![]()
Table 3. Influence of storage period on the concentrations of soluble soil cations.
Means in each row with different letters are significantly different at P < 0.05.
which increased from 3.582 ± 1.306a to 3.968 ± 1.430b. These obtained results were expected specially increasing the storage period logically content in soluble solution. When we compared first and third storage period there were significant difference found in K+ and highly significant difference observed in Mg2+ (from 3.790 ± 2.794a to 7.334 ± 4.739c) and Ca++ (from 11.22 ± 1.978a to 23.505 ± 5.998c) at P < 0.05 level. [15] reported that extractable Ca2+, K+, and Mg2+ were less influenced by storage period, although the effects varied among soil types.
In anions part (Table 4 and Figure 2) respectively, while comparing first and second period results shown that Cl− and ![]() concentrations were significantly increased from0.318 ± 3.553a to 0.749 ± 1.498b and 0.808 ± 3.550a to 0.885 ± 5.710b respectively. However,
concentrations were significantly increased from0.318 ± 3.553a to 0.749 ± 1.498b and 0.808 ± 3.550a to 0.885 ± 5.710b respectively. However, ![]() concentrations were highly significant decreased (from 12.67 ± 2.158a to 3.577 ± 3.186c) with increased of storage period. Similar results were obtained by [15] . Contrasting, [16] reported that soil storage before separation of the soil solution by centrifugation caused a marked decline in P concentrations.
concentrations were highly significant decreased (from 12.67 ± 2.158a to 3.577 ± 3.186c) with increased of storage period. Similar results were obtained by [15] . Contrasting, [16] reported that soil storage before separation of the soil solution by centrifugation caused a marked decline in P concentrations.
3.3. Influence of Temperatures on the Concentrations of Extractable Nutrients
Comparing first and second temperature (10˚C and 25˚C), significant different were found in Mg2+ and Ca2+
![]()
Figure 2. Change in soluble soil anions concentrations at different storage periods.
![]()
Figure 3. Change in soluble anions concentrations at different temperatures.
![]()
Table 4. Influence of storage period on the concentrations of soluble soil anions.
*Means in each row with different letters are significantly different at P < 0.05.
from 3.790 ± 1.452a to 5.568 ± 1.934b and 11.22 ± 1.028a to 17.503 ± 2.103b respectively. Similarly significant different are found while comparing K+ in second and third temperature (25˚C and 45˚C), and highly significant different were found while comparing second and third temperature in Mg2+ and Ca2+ respectively (Table 5 and Figure 3). Similar results were obtained by [15] . Additionally, change in K+ and Ca2+ concentrations during low temperature storage were observed to be less significant and associated with soil types [17] .
Comparing first and second temperature (10˚C and 25˚C), significant different were found in Cl− and ![]() from 0.318 ± 0.185a to 0.749 ± 0.779b and 0.808 ± 1.844a to 0.885 ± 2.967b respectively. [14] , in their study reported that, extractable PO4 of Refrigerated samples always contained lower values than counterparts stored at
from 0.318 ± 0.185a to 0.749 ± 0.779b and 0.808 ± 1.844a to 0.885 ± 2.967b respectively. [14] , in their study reported that, extractable PO4 of Refrigerated samples always contained lower values than counterparts stored at
room temperature. They also pointed out that, extractable ![]() were significantly differences among the soil types. Moreover, the amounts of extractable
were significantly differences among the soil types. Moreover, the amounts of extractable ![]() were significantly increased during low temperature storage [15] . In contrast, [17] , reported that soil storage before separation of the soil solution by centrifugation caused a marked decline in P concentrations, including those of molybdate-reactive P and dissolved organic P, within a few days. However,
were significantly increased during low temperature storage [15] . In contrast, [17] , reported that soil storage before separation of the soil solution by centrifugation caused a marked decline in P concentrations, including those of molybdate-reactive P and dissolved organic P, within a few days. However, ![]() concentrations were significantly influenced by increased temperature (Table 6 and Figure 4).
concentrations were significantly influenced by increased temperature (Table 6 and Figure 4).
4. Conclusion
Depending on our results, all investigated cations and anions were clearly affected by storage periods at various temperatures, exception of Na+. Most cations and anions were increased significantly with increased of storage period and temperatures, while some anions (such as ![]() and Cl−) were dramatically decreased with increased of storage period and temperatures. Despite the simplicity of this research and some limitations such as design which was performed, but the obtained results confirmed that the storage periods and temperatures were influenced markedly on the concentrations of different extractable inorganic soil nutrients. We therefore highly
and Cl−) were dramatically decreased with increased of storage period and temperatures. Despite the simplicity of this research and some limitations such as design which was performed, but the obtained results confirmed that the storage periods and temperatures were influenced markedly on the concentrations of different extractable inorganic soil nutrients. We therefore highly
![]()
Figure 4. Change in soluble anions concentrations at different temperatures.
![]()
Table 5. Influence of storage period on the concentrations of soluble soil anions.
*Means in each row with different letters are significantly different at P < 0.05.
![]()
Table 6. Influence of temperature on different concentrations of soluble anions.
*Means in each row with different letters are significantly different at P < 0.05.
*Corresponding author.
Acknowledgements
The authors are greatly indebted to dean of Agriculture College and general manager of Educational Farm, Nile Valley University.
NOTES
![]()
*Corresponding author.