Received 2 October 2015; accepted 21 February 2016; published 24 February 2016

1. Introduction
The current trend in energy sectors shows a constant dependence on fossil fuel as a transportation energy source. Fossil fuels are non-renewable forms of energy. Relatively recent reports have suggested that global oil and gas reserves would be exhausted in 41 and 63 years respectively [1] . The constant rise in the price of fossil fuels is a serious threat for countries with limited resources [1] and the continuing dependence on fossil fuels as a major source of energy is now recognized to be unsustainable [2] . Exploitation and use of fossil fuel have been suggested as the major driving force behind environmental degradation and associated global climate change [3] . On local levels, the plethora of oil spill over the last five years serve as reminders of the dangers associated with fossil fuel exploitation and use. For example, the 2010 Deepwater Horizon Oil Spill in the Gulf of Mexico is the largest oil spill in US history [4] -[6] , resulting in serious environmental pollution affecting marine and coastal environments. Furthermore, the environmental, economic and societal impacts of such spills last for decades [7] .
Various forms of alternative energy, such as wind, solar, hydroelectric, nuclear, and biofuel are currently being advocated as means of weaning the world’s populations from fossil fuels. However, some of these alternatives come with uncertainties and practical limitations [1] . Among the alternatives mentioned above, biofuels have been receiving the most coverage. Not only can biofuels replace fossil fuels and thereby strengthen the energy budgets of nations, but also, potentials exist for doing so in environmentally desirable ways, including reduction of greenhouse gas emissions, improvement of air quality, reduction of soil erosion, and expansion of wildlife habitat [8] [9] .
Terrestrial biofuel feedstocks have been promoted as potential alternative to fossil fuels, however, they have limitations such as low biomass productivity and competition of arable land for food production [10] [11] . Microalgae have been suggested as potential feedstock for fuel production due to several advantages over terrestrial biofuel feedstock; they possess higher photosynthetic efficiencies, potential for higher biomass production, and higher growth rates compared to terrestrial energy crops. In addition, Microalgae biofuel production has a lower impact on the environment and on the global food supply [11] [12] .
However, cost effectiveness is a major limitation to competitive biofuel production from microalgae [13] . A requirement of a large of water is a major cost during microalgae biomass production. It is estimated that up to 11 - 13 million L∙ha−1∙year−1 is expended during microalgae cultivation, which takes up 10% - 20% of the total cost of production.
In the current emphasis on sustainability, combining algae biomass production with wastewater treatment can reduce costs in algae-oriented biofuel industry [12] [14] , since wastewater contains nitrogen (N) and phosphorus that makes effective growth medium for algal cultivation [8] . Microalgae cultivation using wastewater is now regarded as practical approach for wastewater treatment with simultaneous production of precursors of biodiesel [15] .
In this study, heterotrophic growth rates and biomass yields of the microalgae, C. vulgaris and S. obliquus were assessed in filter-sterilized defined medium (BG-11) and municipal wastewater containing different concentrations of N to determine optimal levels of the nutrient for simultaneous biomass production and wastewater treatment potential the isolates.
2. Material and Methods
2.1. Wastewater Sample Collection
Wastewater was collected from Whites Creek Wastewater Treatment Plant, Nashville, Tennessee, USA to assess growth and biomass production in the medium. The wastewater was allowed to settle, decanted and stored in plastic containers in the refrigerator at 4˚C until used. Initial levels of N and P in the wastewater effluent were determined as 14 and 6.7 mg/L, using Hach DR 890 colorimeter.
2.2. Microalgae Strains and Culture Medium for Pre-Inoculation
For these experiments, Chlorella vulgaris (ATCC® 9765™) and Scenedesmus obliquus (Ankistrodesmus braunii ATCC® 11457™) were obtained from America type culture collection (ATCC). Pre-inoculation cultures were separately grown in 125 mL Erlenmeyer flasks containing 50 mL modified BG 11 medium supplemented with 10 g/L glucose with agitation at 150 rpm under ambient laboratory conditions in 14:10 hour light-dark cycle for 3 days. The modified BG11 medium contained the following components (g/L): The medium consists of the following components; NaNO3 (1.5 g∙L−1), K2HPO4 0.04 (g∙L−1), MgSO4∙7H2O (0.075 g∙L−1), CaCl2∙2H2O (0.036 g∙L−1), citric acid (0.01 g∙L−1), ferric ammonium citrate (0.006 g∙L−1), Na2EDTA (0.001 g∙L−1), Na2CO3 (0.02 g∙L−1) and 1 mL of trace metal solution per liter. The trace metal solution contains a stock of H3BO3 (2.86 g∙L−1), MnCl2∙H2O (1.81 g∙L−1), ZnSO4∙H2O (0.222 g∙L−1), CuSO4∙5H2O (0.079 g∙L−1), and Co(NO3)2∙6H2O (0.0494 g∙L−1) [16] . The pH of the medium was adjusted to 7.5 and it was filtered-sterilized using 0.22 µ filter system.
2.3. Culture Conditions
2.3.1. Growth in Defined Medium
Experiments were performed in 250 mL Erlenmeyer flasks, each containing 100 mL of BG 11 medium with different concentration of N, namely (mg/L): 0.25, 0.50 and 1.00. At the beginning of each experiment, 2 ml of 2% concentration (Vinoculum/Vmedia) microalgal suspension of each microalga species were aseptically transferred to 250 mL flasks. All the 250 flasks were incubated on a rotary shaker at 150 rpm for up to 9 days at room temperature. Microalgal growth was monitored by measuring the optical density of algal suspension daily using DU 730 Life Science UV/VIS spectrophotometer at wavelength of 675 nm. Experiments were carried out in triplicate.
2.3.2. Growth in Wastewater
Wastewater sample was fortified with N as described in above and respective treatments were filtered-sterilized using 0.22 µm filter system to remove microorganisms and fine suspended particles.
2.4. Specific Growth Rate
The specific growth rate (µ) was determined as
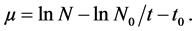 (1)
(1)
where N0 and N represent the optical density at the beginning and any given time t, respectively. A plot of lnN against t yields a straight line, where k was determined through linear regression using the above Equation (1).
2.5. Dry Weight
The dry weights of the three isolates were determined by filtering 30 mL of the culture suspensions on through 90 mm diameter Glass microfiber (GF/B, Whatman). The filters were oven dried at 90˚C to constant weight in a desiccator. The dry biomass was calculated gravimetrically. The algal biomass was expressed in terms of the dry cell weight (DCW) per liter (g∙L−1).
3. Results and Discussion
3.1. Effect of Different Levels of Nitrogen on Growth and Biomass Yield of C. vulgaris in Defined Medium (BG-11)
The overall goal of these investigations is to couple microalgal biomass production to wastewater treatment and to convert biomass produced in the process to precursors of biodiesel. This research recognizes the long-standing knowledge that while production of fuel from microalgae is technically feasible, it is not cost-effective as a standalone process [17] . Rather, investigators have identified wastewater treatment and CO2 mitigation as feasible and highly desirable processes for coupling microalgal biomass production to environmental protection and mitigation with simultaneous biodiesel production [18] .
Furthermore, research has demonstrated that nitrogen metabolism is a critical aspect in biomass productivity of microalgal lipids. We conducted a preliminary assessment of microalgal biomass production in secondary wastewater from Whites Creek Wastewater Treatment Plant TN and found that it contained levels of N that did not support significant biomass productivity for conversion into biofuel. Accordingly, we focused our attention on finding the levels of nitrogen at which growth and biomass productivities of C. vulgaris and S. obliquuscould be optimized in the wastewater. As reference, we compared biomass productivities in standard defined microalgal medium, BG-11 to those in municipal wastewater. Figure 2 shows a plot of optical densities (OD675) over time for C. vulgaris cultivated in BG-11 under varying concentration of N observed over nine days. The plot showed the characteristic sigmoidal growth pattern with different OD optima followed by relatively rapid declines. The 1.0 mg N/L had a highest OD of 9.85 at day 7 compared to 0.50 mg N/L (7.70). There was a decline in OD after 9 days incubation except in 0.25 N/L, which decline at day 8, and increases at day 9. The specific growth rate and biomass productivity of C. vulgaris in BG-11 cultivated in 0.25 mg N/L were 0.28 day−1 and 3.50 g∙L−1 respectively. Increasing levels of N to 0.50 mg/L showed an increase in growth rate and biomass yield to 0.37 day−1 and 4.28 g∙L−1 respectively. A further increase of N to 1 mg/L caused an increase in growth rate to 0.43 day−1. However, at this level of N addition, biomass yield decreased to 3.64 g∙L−1 (Figure 4). A similar study was carried out in 100% piggery wastewater reported that the maximum specific growth rate and biomass productivity of microalga obtained were 0.839 d−1 and 0.681 g∙L−1∙d−1, respectively [21] . Another study showed that in a semi-continuous cultivation of microalgae, the specific growth rate and biomass of Chlorella sp. cultures in the conditions aerated with 2% - 15% CO2 were 0.58 - 0.66 d−1 and 0.76 - 0.87 g∙L−1, respectively, and the rate of CO2 reduction was up to 58% [19] . Previous studies have reported at lower nitrogen (<5 mg/L) microalgal lipid content oil can be as high as 40% supporting the fact that biomass productivity is proportional to nitrogen depletion [20] . Our findings also supported previous study that reported the nitrogen sequence of nitrate conversion to nitrite and then ammonium before been incorporated into microalgal metabolic pathway enhance biomass productivity through its nitrogen deficiency [20] .
3.2. Effect of Different Levels of Nitrogen on Growth and Biomass Yield of C. vulgaris in Wastewater
The isolate C. vulgaris grown in wastewater containing different levels of 0.25, 0.50 and 1.0 mg N/L were able to survive in the wastewater used. The main reasons for coupling microalga biodiesel production with waste- water treatment are to reduce the dependence on freshwater, minimize the cost of nutrients used in algal cultivation and to eliminate N and phosphorus from wastewater [22] . The specific growth rates were 0.44, 0.32, and 0.32 day−1, respectively for cultures fortified with 0.25, 0.50 and 1.0 mg N/L, (Figure 1). Their corresponding biomass productivities were 4.96, 2.19, and 2.31 g∙L−1, respectively (Figure 4). The growth rates of microalgae are often lower in various industrial wastewaters due to low nitrate and phosphate and high toxin levels [8] . The fortified wastewater used in this study was able to support the microalgal growth. Interestingly, the addition of 0.25 mg N/L to wastewater of C. vulgaris showed the highest OD (8.99 as shown in Figure 2) at day 8 with a biomass
![]()
Figure 1. Specific growth rate of C. vulgaris and S. obliquus in defined medium and municipal wastewater.
![]()
Figure 2. Growth curves Chlorella vulgaris cultivated in batch experiment on a rotary shaker at room temperature and 150 rpm under heterotrophic condition for 9 days.
yield of 4.96 g∙L−1. This indicated that the organism performed better at 0.25 mg N/L than wastewater supplemented with 0.50 and 1.0 mg N/L. It demonstrated that the biomass productivity increased with the decreased concentration of nitrogen. Since, the main reason for combining microalga biomass production with wastewater treatment is to reduce the dependence on freshwater and minimize the cost of nutrients used in algal cultivation. A similar study conducted using Chlamydomonas obtained the maximal biomass of 1.5 g∙L−1 after 5 days of cultivation [22] .
3.3. Effect of Different Levels of Nitrogen on Growth and Biomass Yield of S. obliquus in Defined Medium
In this study, S. obliquus was also evaluated under the same conditions described for C. vulgaris. The optical density (Figure 3) monitored over a period of 9 days showed a similar pattern to the biomass production. The OD of 0.25 mg N/L reached the highest OD (6.74) at day 9. Scenedesmus obliquus grown in BG-11 with N additions of 0.25, 0.50 and 1.0 mg/L were 0.59, 0.73 and 0.71 day−1, respectively (Figure 1). BG-11medium containing 0.50 mg N/L had a higher growth rate of 0.73 day−1. Correspondingly, to different growth rates observed in BG-11, biomass productivities by S. obliquus were 2.15, 3.10 and 3.37 g∙L−1, respectively (Figure 4).
![]()
Figure 3. Growth curves Scenedesmus obliquus cultivated in batch experiment on a rotary shaker at room temperature and 150 rpm under heterotrophic condition for 9 days.
3.4. Effect of Different Levels of Nitrogen on Growth and Biomass Yield of S. obliquus in Wastewater
As shown in Figure 1 and Figure 3, the specific growth rate of 0.25, 0.50, 1.0 mg N/L W, were 0.56, 0.47, and 0.55, respectively. The corresponding biomass productivities were 1.05 ± 0.0.16, 1.18 ± 0.08 and 1.27 ± 0.06 g∙L−1 respectively. However, this is in contrast to higher specific growth rate and biomass yield observed in wastewater for C. vulgaris. Among the six microalga isolates investigated during a similar study, the average specific dry biomasses of 0.53 and 0.49 g/dwt/L were found for S. obliquus, and C. vulgaris, respectively, after 20 days of cultivation in piggery wastewater [23] . In another study that evaluated the prospect of using microalgae S. obliquus for brewery wastewater treatment and biomass production. After 9 days, a maximum of 0.9 g∙L−1 was observed at 12 h period of daily light exposure in aerated culture [24] .
3.5. Comparison of Biomass Production between C. vulgaris and S. obliquus
Comparing the graph of Figure 4, the biomass productivity of C. vulgaris was generally higher than the productivity of S. obliquus under varying conditions of the test media. The biomass productivity of C. vulgaris was significantly higher than S. obliquus in 0.25 N/L BG-11, 0.50 N/L BG-11, 0.25 mg N/L W, 0.50 mg N/L W, and 1.0 mg N/L W, respectively at p < 0.05. The biomass productivity of C. vulgaris in 0.25 mg N/L W was significantly
![]()
Figure 4. Biomass of Chlorella vulgaris and Scenedesmus obliquss dried to a constant weight (90˚C) overnight.
higher than the production in 0.50 mg N/L W and 1 mg N/L W. A higher significant productivity of S. obliquus was observed in 0.50 mg N/L BG 11 as compared with 0.25 mg N/L W, 0.50 mg N/L W, and 1 mg N/L W. The biomass yield increased as the N concentration was increased from 0.25 mg to 0.50 mg N/L, a similar study conducted showed the highest biomass obtained was 10.04 g∙L−1 with biomass productivity of 1.673 g/L d was obtained using 1.0 g∙L−1 of NaNO3 as N source and 40 g∙L−1 glucose, while a biomass of 7.09, 8.55 and 9.45 g∙L−1 with biomass productivity of 1.182, 1.425 and 1.575 g/L d were found at 20, 30 and 50 g∙L−1 glucose, respectively [25] . In another study, different pretreatment options test and filtration (by 0.2 μM) resulted in the highest biomass and lipid productivity [26] . The effect of N concentration on of Chlorella sp. showed that lower N supplement 0.50 mg N/L BG 11 and 0.25 mg N/L W, increased the biomass yield to 4.28 ± 0.22 and 4.96 ± 0.17 g∙L−1, respectively, when compared to 1mg N/L BG 11 (3.64 ± 0.68 g∙L−1). The results obtained in this research have similar trend with the result that showed excess N source reduces productivity of C. vulgaris [27] . The highest biomass productivity was obtained at 10 mM sodium nitrate, with a biomass concentration of 3.2 g∙L−1 and a biomass productivity of 0.63 g−1∙day−1 [28] .
4. Conclusion
In this study, C. vulgaris cultivated in 0.25 mg/L N wastewater showed the most significant increases to growth rate and biomass productivity. Overall, growth and biomass productivities of S. obliquus showed that, this isolate grew slower and produced lower biomass than C. vulgaris under all varying conditions. Based on high biomass productivity of C. vulgaris, this result suggests that it could be a potential candidate for further consideration for wastewater treatment and biofuel production. Further studies would determine rates and magnitudes of nitrogen and phosphorus assimilation and lipid productivities of the isolates. Studies are elucidating metabolic processes under which lipid accumulations are optimized by the isolates, including, but not limited to, nitrogen starvation in cultures. Using microalgae for wastewater treatment offers significant advantages over conventional wastewater treatment, since microalgae utilize inorganic nutrients present in wastewater for their metabolism. Coupling the high biomass productivity of wastewater-grown microalgae for biofuel production offers real opportunities for sustainable and renewable energy and environmental enhancement.
Acknowledgements
The authors gratefully acknowledge staffs of Whites Creek Wastewater Treatment Plant, Nashville, Tennessee, USA for providing wastewater samples and Department of Agricultural Sciences, Tennessee State University for funding support. We thank Tennessee State University students, Shola Awoyemi and Oluwatosin Ogunma- yowa, for assistance in data analyses and review.
NOTES

*Corresponding author.