Prevalence and Factors Associated with Anaemia among Pregnant Women Attending Antenatal Clinic in the Second and Third Trimesters at Pumwani Maternity Hospital, Kenya ()
Received 24 November 2015; accepted 16 January 2016; published 19 January 2016

1. Introduction
Anaemia, defined as a decreased concentration of blood hemoglobin, is one of the most common nutritional deficiency diseases observed globally and affects more than a quarter of the world’s population [1] . It is estimated that 41.8% of pregnant women worldwide are anaemic. In Africa, 57.1% of pregnant women are anaemic [2] . In Kenya, the prevalence of anaemia among pregnant women is 55.1% [3] . When the prevalence of anaemia among pregnant women is 40.0% or more, it is considered as a severe public health problem [4] . Anaemia during pregnancy is considered severe when Hb concentration is less than 7.0 g/dl, moderate when Hb level is 7.0 - 9.9 g/dl, and mild when Hb level is 10.0 - 10.9 g/dl [5] -[7] . Anaemia is a major cause of morbidity and mortality of pregnant women and increases the risks of foetal, neonatal and infant mortality [8] . Anaemia during pregnancy contributes to 20% of all maternal deaths [9] .
In developing countries, the cause of anaemia during pregnancy is multifactorial and includes nutritional deficiencies of iron, folate, and vitamin B12 and also parasitic diseases, such as malaria and hookworm. Iron deficiency is the cause of 75% of anaemia cases during pregnancy [5] [10] . HIV infection is also implicated with high prevalence of anaemia in sub-Saharan Africa [11] [12] . Iron deficiency anaemia affects the development of the nation by decreasing the cognitive and motor development of children and productivity of adults [5] [13] . Despite its known effect on the population, the available data regarding the determinants of anaemia during pregnancy in Kenya and particularly in Nairobi are limited. Hence, this study was aimed at determining the prevalence and factors associated with anaemia among pregnant women attending ANC at PMH, Nairobi, Kenya.
2. Methods
2.1. Study Setting
The study was conducted at Pumwani Maternity Hospital which is a referral maternity hospital located on the East of Nairobi City (country’s capital city). The hospital is a pioneer in the provision of maternity care in sub-Saharan Africa. It has 354 obstetric beds, 144 baby cots and 2 theatres. Daily normal deliveries are 50 - 100, and Caesarean Sections are 10 - 15. To date the Hospital remains the largest maternity hospital in the country and sub-Saharan Africa. It is equivalent to a Provincial Hospital in status and is reported to be third busiest maternity hospital in African.
2.2. Study Design and Participants
A hospital based cross-sectional study was conducted from 8th June to 18th August, 2015. The study population included in this study was all 2nd and 3rd trimester pregnant women attending ANC at Pumwani Maternity Hospital.
2.3. Sample Size Determination
The sample size was determined by using single population proportion formula (Fischer et al., 1998; n = Z²pq/d²) by considering 95% CI, 55.1% proportion of anaemia during pregnancy from the study carried out by the Ministry of Health, Kenya in 2013 and absolute precision of 0.05 which gives a sample size of 380. However, since the population during the study period was below 10,000, sample size adjustment was done using the following
formula, 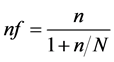 resulted in 258 subjects.
resulted in 258 subjects.
where:
nf = The desired sample size (when the study population is less than 10,000);
N = Total population (around 800 antenatal mothers, in the 2nd and 3rd trimesters in 10 weeks period);
n = The desired sample size (when the study population is more than 10,000) which is 380.
2.4. Sampling Method
A review of ANC attendance register showed that about 16 pregnant women in the second and third trimester attend ANC at Pumwani Maternity Hospital daily. Accordingly the study was designed to be performed for 10 weeks during which period an estimated 800 women in second and third trimester were expected to visit for antenatal care. Systematic random sampling method was used to select study participants. The estimated number of 800 pregnant women (2nd and 3rd trimester) in 10 weeks period was divided by the minimum adjusted sample size (258) to give the sampling interval of 3. The first pregnant woman included in the sample was chosen randomly by blindly picking one of three pieces of paper named for the first three visitors. After that, every third pregnant woman who came for antenatal care was recruited in the study until the desired sample size was attained. To avoid double participant recruitment, the research assistants confirmed whether the participants had previously visited the ANC at the hospital and participated in the study during the data collection period.
2.5. Data Collection
A pre-tested semi-structured questionnaire was used to collect data by trained research assistants. Data regarding participants’ demographic and socio-economic characteristics, obstetric and medical history, ANC visits and iron and folic supplementation, dietary habits and anthropometric measurements were obtained. Haemoglobin concentration was measured using portable HEMOCUE B-Hb photometer. One drop of capillary blood via finger prick was used to estimate haemoglobin level.
2.6. Data Analyses
Data were analysed using SPSS Version 20. Descriptive statistics such as frequency, percentage, mean and standard deviation were used to describe selected variables. Pearson’s chi-square test and odds ratio (OR) with corresponding 95% confidence intervals (CI) computed to find association between independent and dependent variable (anaemia). A P-value of <0.05 was considered statistically significant. Variables having a P-value <0.05 in the bivariate analysis were subjected into a multivariate analysis to determine factors independently predicting anaemia during pregnancy.
2.7. Ethical Consideration
Ethical approval was obtained from the University of Nairobi/Kenyatta National Hospital, Ethics and Research Committee. Permission to conduct the study was also obtained from the hospital administration. Written informed consent was obtained from each study participant. Confidentiality was maintained at all levels of the study. Pregnant women who found to be anaemic were advised by the research assistants to seek proper treatment and follow-up.
3. Results
3.1. Socio-Demographic and Economic Characteristics of Respondents
Table 1 shows the socio-demographic and economic characteristics of the pregnant women. The mean age of
![]()
Table 1. Socio-demographic and economic characteristics of respondents.
the women was 26.4 years (SD ± 4.7). Out of the 258 participants, 104 (40.3%) and 102 (39.5%) were in the age group of 18 - 24 years and 25 - 30 years respectively. The remaining 52 (20.2%) were 31 years and above. All of the participants were from Nairobi County. Majority of the women were married (84.9%), employed (67.4%) and Christian followers (90.3%). Regarding educational level of the participants 54 (21%), 121 (46.9%) and 78 (30.3%) attended primary, secondary and tertiary level of education respectively. The highest percentage 127 (49.2%) of the participants had monthly income between 10,000 - 30,000 Ksh. About a quarter 68 (26.4%) of them had monthly income of less than 10,000 Ksh.
3.2. Obstetric History of the Pregnant Women
The obstetric history of the participants is presented in Table 2. The mean gestational age was 28.2 weeks (SD ± 8.1). The majority 165 (64.0%) of the participants were in the third trimester of pregnancy and 93 (36.0%) were in the second trimester of pregnancy. One hundred and fifty one (58.5%) of the women were multigravida while 107 (41.5%) were primigravida. Of the multigravida women 121 (80.1%) had an inter-pregnancy interval of two years or more. The remaining 30 (19.9%) had birth interval of less than two years.
3.3. Ante Natal Clinic (ANC) Attendance and Taking IFAS
Table 3 shows ANC visits and IFAS supplementation during the current pregnancy. Majority (92.2%) of the participants started attending ANC after 12 weeks of gestation. Most 217 (84.1%) of the women attended ANC
![]()
Table 2. Obstetric history of the participants.
![]()
Table 3. Ante natal clinic attendance and taking IFAS.
for the first time between 12 and 24 weeks of their gestational age. Almost half 89 (50.9%) of the women started taking IFAS at 19 - 24 weeks of gestation. Sixty seven (38.3%) of them started taking IFAS at 12 - 18 weeks of their gestation. Only 13 (7.4%) of the women had started taking IFAS before 12 weeks of their gestation.
3.4. Health Conditions of the Women during the Current Pregnancy
Majority 213 (82.6%) of them reported they did not experience any health problem. Very small number 8 (3.1%) and 23 (8.9%) of the women reported they had diarrhoea and febrile illnesses for the last one month preceding the study respectively. Majority of the women were sero-negative while 20 (7.8%) were sero-positive (Table 4).
3.5. Anthropometric Measurements
An assessment of weight, height and MUAC was done for all the pregnant women. Majority169 (65.5%) of the women had above 24.9 body mass index (BMI) and 88 (34.1%) of them had normal BMI (18.5 - 24.9). The mean MUAC for the women was 25.1 cm (SD ± 3.5). About three quarter 187 (72.5%) of the women had MUAC of 23 cm or above and about a quarter 71 (27.5%) of the women had MUAC of less than 23 cm (Table 5).
![]()
Table 4. Health conditions of the women during the current pregnancy.
![]()
Table 5. Anthropometric measurements.
3.6. Life Style Habits
The majority 241 (93.4%) of the women said they drink beverages (tea, cocoa or coffee) and 66.0% of them drink these beverages in less than 20 minutes before/after meals. More than half 143 (55.4%) of the women ate meals three times per day. One third 86 (33.3%) of the women avoided certain foods such as red meat 28 (32.6%) and green vegetables 26 (30.2%) due to vomiting 37 (43.0%) and heart burn 19.8%. A few mothers 51 (19.8%) craved for non-food substances and the common type of craving was stone 44 (86.3%). All of the women reported they never smoke cigarettes and almost all (98.4%) of the women said never taken alcohol (Table 6).
3.7. Prevalence of Anaemia
The cut-off point of Hb concentration for diagnosis of anaemia during pregnancy was taken at 11 g/dl (McLean et al., 2008). The overall prevalence of anaemia (Hb < 11 g/dl) was 57.0% with 95% CI of 51.0% to 63.0%.The mean Hb concentration was 10.32 gram/deciliter (g/dl) with a SD of ±1.72 g/dl. In terms of severity, mild anaemia was 26.5%, moderate anaemia was 70.7% and severe anaemia was 2.7%.
3.8. Factors Associated with Anaemia Using Unadjusted and Adjusted Logistic Regression
In the bivariate analysis, participants’ age, marital status, employment status, iron and folic acid supplementa-
![]()
Table 6. Life style habits of participants.
tion during the current pregnancy, febrile illness, HIV status and nutritional status (MUAC) were significantly associated with anaemia among the pregnant women. However, following multivariate analysis maternal age, employment status, nutritional status (MUAC) and iron and folic acid supplementation during the current pregnancy remained significantly and independently associated with anaemia.
Pregnant women aged 31 years and above were about 3 fold more likely to be anaemic [AOR = 2.71; 95% CI = 1.25 - 5.88; P = 0.012] compared to those aged 18 - 24 years. Government/private employed pregnant women had 2.9 times [AOR = 2.94; 95% CI = 1.47 - 5.88; P = 0.002] and those who were self-employed had 1.9 times [AOR = 1.91; 95% CI = 1.03 - 3.53; P = 0.039] more likely to be anaemic compared to those who were housewives. Pregnant women who did not take iron supplementation during the current pregnancy were 2 times more likely to develop anaemia [AOR = 2.04; 95% CI = 1.14 - 3.64; P = 0.016] than those who took the supplements. Pregnant women with MUAC of less than 23 cm were about 2.5 fold more likely to be anaemic [AOR = 2.52; 95% CI = 1.36 - 4.67; P = 0.003] compared to those who had MUAC of 23 cm and above (Table 7).
![]()
![]()
Table 7. Factors associated with anaemia using unadjusted and adjusted logistic regression.
Abbreviations: COR = Crude Odds Ratio, AOR = Adjusted Odds Ratio, CI = Confidence Interval, *P < 0.05, **P < 0.01, ***P < 0.001.
4. Discussion
The present study showed that the prevalence of anaemia among pregnant women was high at 57%. According to WHO classification of the public health importance of anaemia, it is a severe public health problem [4] . This finding is slightly higher than the `Kenya national prevalence of anaemia in pregnancy at 55.1% [3] . It is also much higher than to the study carried out in Kakamega County (Kenya)reported at 40% [14] and unpublished finding from Mbagathi District Hospital, Nairobi at 36.2% [15] . This variation can be due to the fact that the participants of this study did not include pregnant women in the first trimester as anaemia is more common in the second and third trimesters. However, the figure is relatively comparable to other studies conducted in African countries such as Nigeria at 54.5% [16] and Ethiopia at 56.8% [17] . But it is lower than the findings from Uganda at 63.1% [18] and Egypt at 62.2% [19] and higher than Tanzanian finding at 47.4% [20] .
In the present study the odds of anaemia were observed to rise as maternal age advances. Pregnant women aged 31 years and above were significantly more anaemic compared to those mothers 18 - 24 years. This result is in agreement with the previous studies conducted in Kisumu District of Kenya [21] , Ethiopia [22] , Tanzania [23] and Egypt [24] which found that late pregnancy has significantly increased risk of developing anaemia. It is generally believed that anaemia in pregnancy increases with rising parity and maternal age. Besides the general body weakness with advanced maternal age, older women are expected to be multigravida. Multigravida may induce anaemia by reducing maternal iron reserves at every pregnancy and by causing blood loss at each delivery [25] .
The proportion of anaemia was significantly more among employed participants compared to the housewives. This finding is in line with studies conducted in Pakistan [26] and Brasil [27] . This was not unexpected, where women often need to work outside the home because of low family income. These women could not have enough resting time as a pregnant mother and enough time to attend ANC compared to housewives. Furthermore, most of these women would be considered underemployed, i.e., employed but with jobs that were unsteady and low-paying. This can greatly contribute to the well-being of the women and the positive outcome of the pregnancy.
The odds of developing anaemia was significantly more among pregnant women who did not take iron and folic acid supplements. This finding corroborates with previous studies such as in Ethiopia [17] [28] , Uganda [29] , Nigeria [30] , Vietnam [31] and India [32] which indicated that lack of iron supplementation is among the most significant risk factors for developing anaemia during pregnancy. The reason for this might be pregnant women who take their iron tablets which can help them to increase their hemoglobin level and prevent anaemia during pregnancy time. Even if the woman had a normal amount of iron before pregnancy, more is needed during pregnancy due to increased demand. This requirement is not met by food alone in developing countries like Kenya, and therefore oral iron supplementation is justified [16] .
Pregnant women with MUAC of less than 23 cm had higher prevalence of anaemia. This finding is consistent with previous studies in Western Kenya [33] , Ethiopia [17] , India [34] and Nepal [35] , which found that MUAC of less than 23 cm significantly increase the risk of developing anaemia. This can be explained by the fact that undernourished pregnant women have a higher probability of being deficient of micronutrients and therefore more likely to develop anaemia. Pregnancy is the most nutritionally demanding period in a woman’s life. Consequently, pregnant women are advised to eat more diversified diet than usual [36] .
5. Limitation of Study
One of the limitations of this study is the nature of the study design, being as a cross-sectional study design, it does not show which preceded anaemia or the risk factors. This limits measuring the cause and effect relationship. Second, it did not classify the types of anaemia based on red blood cell morphology which would give clear picture on the types of anaemia. Third, as the current study was conducted entirely within one hospital and all the participants were from Nairobi County, differences of geographic location were not assessed. Therefore, generalizability to other hospitals and rural areas in the country may not be possible.
6. Conclusion
Anaemia is found to be a severe public health problem in the study area. Late pregnancy, employment, not taking IFAS during the current pregnancy and MUAC of less than 23 cm were significantly and independently associated with anaemia during pregnancy. Special care should be given during late pregnancy and for employed pregnant women to prevent anaemia. Nutritional counseling on consumption of more diversified and extra meal and iron-rich foods and iron/foliate supplementation are recommended to prevent anemia in pregnant women. Further research (more robust) on risk factors of anaemia during pregnancy including urban and rural communities should be carried out to strengthen and broaden these findings.
Acknowledgements
The authors are grateful for all study participants who took part in the study for their time. We also acknowledge the National Board for Higher Education of Eritrea and African Development Bank for funding the study.
Competing Interest
The authors declared that they have no competing interests.
Abbreviations
ANC: Antenatal care; AOR: Adjusted odds ratio; CI: Confidence interval; COD: Crude odds ratio; Hgb: Hemoglobin; IDA: Iron deficiency anemia; MUAC: Middle upper arm circumference; OR: Odds ratio; SPSS: Statistical package for social science; WHO: World Health Organization.
NOTES

*Corresponding author.