Received 14 December 2015; accepted 20 February 2016; published 23 February 2016

1. Introduction
Brinjal (Solanum melongena L.) is one of the most important vegetable crops of Andaman and Nicobar Islands. These islands are located about 1200 km from mainland India and have altogether different environmental regime including, soil, precipitation, water and farming practices. Bacterial wilt of Brinjal caused by Ralstonia solanacearum is a serious disease, which limits eggplant production from 4.24 to 86.14 per cent [1] while in islands the disease is devastating in humid and congenial climate and can cause up to 100% losses. Bacterial wilt is one of the most important diseases of solanaceous crops including brinjal in these islands. Recently, this disease has risen to alarming proportion in the plains of India. Many of the commercial varieties are growing but they are susceptible to this disease [2] . Unfortunately, the majority of resistant varieties developed in mainland India do not survive Island wilt conditions. The resistance reactions and mechanism of resistance are very location and environment specific. Genetic behaviour of bacterial wilt can be depicted from the segregating populations. The dominant and recessive major gene or multiple gene actions help us to decide upon the suitable strategy to develop a resistant variety or hybrid as well as the suitability of donor sources and recurrent parents. A good donor is the line which offers minimum linkage drag, easily maintained and cross compatible and good combining ability. Mechanisms of resistance are also very important to be understood before utilizing the source for any breeding programme. Plants can exhibit immune, resistant, escape, stage specific resistance or a combination of multiple factors in additive or epistatic interaction mode. The resistance reactions and mechanism of resistance are very location and environment specific [3] and the very little success has been attained using chemical control measures [4] . The agency of R genes present in plants if identified then designing of breeding lines becomes a better choice. Therefore, development of resistant varieties suitable for islands is most economic, eco-friendly and feasible method to ensure better productivity of brinjal. Therefore, the present study was undertaken to understand the genetic behaviour of bacterial wilt of brinjal in tropical islands conditions.
2. Material and Methods
For initial idea the brinjal entries from mainland India were screened under sick plot conditions having higher bacterial load (1 × 107 cfu). Seedlings of each entry were raised in sterile soil media without any biotic stress. The one month old seedlings were transplanted in sick plot for evaluation of disease reaction. The “CARI-B-1” is resistant to bacterial wilt and bear green oblong fruits [5] while “PPL” is a susceptible parent for bacterial wilt and produce purple fruits in clusters (AICRP-VC, 2012).
The segregating population were developed by selfing of single F1 plant and resistant plants of F2 were used for development of F2-3 families. A set of 42 plants from each F2 surviving plants were used for observations in sick plot having sufficient inoculum load of (1 × 107 cfu) of the pathogen. The segregating population was raised in close spacing (60 × 40 cm) with raised with proper package of practices as described by [6] . The observations were recorded on bacterial wilt incidence on fortnight basis. The percent disease incidence was recorded by
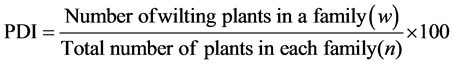 . The progeny families were then plotted against percent
. The progeny families were then plotted against percent
disease incidence on given fortnight intervals order to understand the progressive disease development pattern in the form of regression equation and R2 values were taken as best estimate of regression model. The genetic ratios were tested based upon goodness of fit for inheritance using chi-square test.
3. Results
3.1. Disease Reaction in Mainland Entries
30 lines of cultivated brinjal (Solanum melongena) obtained from mainland All India Coordinated Research Project (AICRP) on vegetable crops were tested against wilt incidence under sick plot conditions in research farm of Central Island Agricultural Research Institute, Port Blair, which is situated in South Andaman. The bacterial wilt (BW) incidence ranged from 11.67% - 96.67% with mean value of 60%. None of the line showed full resistance to bacterial wilt, however line BRBWRES1 showed better performance as compared to check and other lines with only 11.67% disease incidence during first two months however, later on this also collapsed with exponential expression of bacterial wilt.
3.2. Disease Reaction in Biparental Segregating Population
A biparental segregating population from (“CARI Brinjal 1” × “Pusa Purple Long”) cross was developed. From single F1 fruit the (F2) population comprising of 67 plants was developed. Out of 67 plants only 22 plants survived up to 60 days. This suggested a simple fitting ratio of 1:3 showing recessive major gene action (Table 1). Out of these 22 plants five plants did not bear any flower and fruit and one typical character of these five plants was that these were highly pigmented. Only 17 could be taken to next level (F3) due to problem in fruiting of five plants. However, further dissection of the resistance in F3 progeny families indicated towards the complex nature of bacterial wilt genetic behaviour. A total of 17 F3 progeny families each comprising 42 plants in fruit to row manner were evaluated in bacterial wilt sick plot along with two replicated checks viz. Check 1 (Diglipur local ) and Check 2 (CARI Brinjal 1) The disease reaction was recorded at fortnightly intervals. Family of plants derived from ANBF3-11-57 progeny showed no mortality up to fifth fortnight and mortality reached 33% in this group at the end of sixth fortnight. A polynomial regression equation of order 4 was best fitted to this family with coefficient of determination R2 value of 0.996. ANBF3-11-16 and ANBF3-11-62 progenies behaved like Check 2 exhibited an initial mortality of less than 20% up to fourth fortnight and then slight increase in fifth and sixth fortnight reaching to 31%. These two progeny families depict a typical resistant category following an exponential regression trend line with R2 value of 0.946.
Three progeny families viz. ANBF3-11-04, ANBF3-11-11 and ANBF3-11-20, followed Check 1 (Diglipur local) with an exponential regression trend line with R2 value of 0.944 which showed breakdown of resistance during 5th and 6th fortnights suggesting that such plants can show moderate level of resistance for economic harvest of the crop. It also indicates that factor for inducing resistance at later stage are lacking in these lines and escape mechanism is operating. ANBF3-11-29, ANBF3-11-50 and ANBF3-11-64 exhibited a linear trend with R2 value of 0.986 and moderate susceptibility. The regression trend line of five progeny families’ viz. ANBF3- 11-03, ANBF3-11-09, ANBF3-11-27, ANBF3-11-30, and ANBF3-11-41 were also found to exhibit linear progression of disease development with a greater slope indicating higher susceptibility. The progeny families ANBF3-11-47, ANBF3-11-49 and ANBF3-11-67 showed a sudden breakdown of resistance in second and subsequent fortnights and these families fall in highly susceptible category following a high order polynomial regression equation with R2 values of 0.989 to 0.996. This analysis indicate that though the resistance against bacterial wilt is governed by recessive gene action yet there preponderance of complex genic interactions due as revealed by the regression analysis. Another interpretation of results indicate that if we classify all the families in two categories of resistant and susceptible keeping resistant bar up to 40% mortality and consider duration of observations up to 75 days after transplanting than it fits well in goodness with 1:3 category though it seems to be convenient for backing up the previous findings, however it is ambiguous and contradictory to the fact that there is existence of two classes in resistant category and three classes in susceptible category wherein at fifty percent plants are surviving up to fifth fortnight in one category giving some yield and there is 50% mortality in fourth fortnight which is peak period of reproductive stage of plant and the third category which is dying in vegetative stage itself (Table 2). The mechanism of resistance is shown by the progeny families which show a parallel line with time Figure 1(a) and Figure 1(b). Few entries also express early resistance and then breakdown which help in escape Figure 1(c) while the third category shows progressive escape with slow mortality Figure 1(d). The Fourth category is of progressive susceptibility with time as the inoculum multiplies in the rhizosphere Figure 1(e) while the fifth category shows a sudden breakdown at early stage itself suggesting highly susceptible reaction Figure 1(f).
![]()
Table 1. Chi-square values of different segregating populations.
![]()
Figure 1. Resistance mechanisms prevalent in segregating population. (a) and (b) resistance mechanism (Resistance reaction exhibited by three families and CARI Brinjal-1 as check2); (c) moderately resistant with Early Resistance + escape mechanism; (d) progressive linear susceptibility; (e) susceptible; (f) highly susceptible. Note: X-axis is the fortnightly (15 days) interval and Y-axis depicts the percent disease incidence.
This indicates there is more than one gene involved in governing stage specific resistance or there is agency of modifiers/transcription factors which result in differential expression of resistance action over the time. In total five categories of resistance reactions were observed in F3 progenies. It seems to be an additive effect of dif-
![]()
Table 2. Percent disease incidence in progeny families.
ferent genes comprising a gene family which impart resistance at different levels of plant development and absence of allele causes stage specific susceptibility.
Similar effects have been observed in another segregating population (F2) of a local susceptible accession of cultivated brinjal (S. melongena) and wild relative S. torvum which is highly resistant to all kinds of wilts. The F2 progeny raised from the single fruit of F1 plant comprising of 110 plants. A set of 40 plants was inoculated with the Ralstonia solanecearum (1 × 108 cfu) after 21 days of transplanting. 31 plants died within 30 days after inoculation while 9 plants survived suggesting a recessive inheritance of bacterial wilt. However, the mortality of plants was not sudden rather it happened over a period of time. When we plot the results on line diagram and observe we can see the different categories of resistance reaction indicating the mechanisms of resistance prevalent in the segregating population.
4. Discussion
The study of inheritance pattern of resistance to many biotic stresses is helps the plant breeders to device suitable methods to develop resistant hybrid/variety. The bacterial wilt inheritance is race, biovar, strain, genotype and environment specific and it is genetically determined. There are different schools of thought over the number of genes and dominance behaviour of resistance. Many workers have reported the resistance to be governed by single dominant gene [2] [7] - [9] and they have also mentioned that single gene inheritance for bacterial wilt resistance. Swaminathan and Srinivasan [8] studied the resistance to P. solanacearum in the hybridization between eggplant cultivar purple long Dutta and Solanum melongena var. insanum and reported the latter carried a single dominant gene for resistance and purple long Dutta carried a recessive gene. Vijayagopal and Sethumadhavan [10] confirmed the above study by attempting intervarietal hybrids of the same cross i.e., Solanum melongena Pusa Dutta × Solanum melongena var. insanum and reported resistance to Pseudomonas solanacearum was dominant and monogenic controlled. Resistance to bacterial wilt was inherited as single dominant gene character in the cross WCGR-112-8 and Pusa kranti in brinjal [2] which was also the case of the resistance to bacterial wilt in “Nihan nassu” cultivar was due to the presence of single dominant gene [11] . Study on the 11 hybrid families from Solanum phureja × Solanum tuberosum crosses indicated that, resistance was controlled by three dominant independent genes [12] . Chaudhary [13] reported that the bacterial wilt was controlled by polygenes. The inheritance pattern for bacterial wilt resistance has been reported ranging from monogenic dominant/ recessive to inhibitory type in different cross combinations by different workers. Feng et al. [14] concluded that the inheritance of bacterial wilt resistance as additive-dominant model. Additive, dominant and recessive effects played major role in the genetics of bacterial wilt resistance, with additive effects being the dominant factor. Bacterial wilt resistance was controlled by number of minor, major and cytoplasmic genes. However, the current study supports inheritance study of susceptibility to bacterial wilt was dominant over resistance in brinjal [15] .
Acknowledgements
The authors acknowledge the active support of field and laboratory staff. Authors also acknowledge the support of Director, CIARI for providing suitable funding and resources for conducting this study.
NOTES
![]()
*Corresponding author.