Simultaneous Removal of Chromium and Lead from Water by Sorption on Iraqi Montmorillonite ()
1. Introduction
Chromium and lead are among the toxic heavy metals in the environment. Lead contamination comes mainly from lead in paint, gasoline, water distribution systems, food, and lead used in hobby activities. Lead exposure attributable to automobile air emissions was a major exposure source prior to 1976. The use of leaded gasoline declined in developed countries but was still used in other countries. Water is contaminated in the distribution networks and the possibility of contamination will increase with the decrease of pH of water. When lead-paint is deteriorated, the risks of lead poisoning increase to children because of abrasion into dust. Food may also be contaminated by the lead through the soil contaminated with lead or the use of insecticides that contain lead [1] . Lead exposure may cause renal disease, irreversible neurologic damage and cardiovascular effects. Lead in blood increases dramatically in the people who work in crowded intersections when leaded gasoline is still in use. Lead will move to living bodies by grazing and food chains. A recent study indicates that the wire and cable manufacturing and the rubber and plastic industry are the largest reported users of lead compounds [2] .
Many techniques were utilized for the removal of lead from aqueous medium like emulsion liquid membrane [3] , adsorption on bagasse ash [4] , adsorption on clay [5] , biosurfactant, [6] , and ion-exchange with faujasite tuff [7] .
Several materials and methods were used for the removal of chromium like precipitation [8] [9] , ion exchange [10] [11] , and wastewater sludge [12] . Adsorption was widely used for the removal of chromium using waste ma- terials [13] - [16] . Infiltration in soil, and uptake by wetland plants, also accounted for some removal practices [17] .
The main human activities that increase the concentrations of chromium are steel, leather, textile manufacturing and electro painting. These applications will mainly increase concentrations of chromium in water. Through coal combustion chromium will also end up in air and through waste disposal chromium will end up in soils. Most of the chromium in air will eventually settle and end up in waters or soils. Chromium in soils strongly attaches to soil particles and as a result it will not move towards groundwater. In water, chromium will absorb on sediment and become immobile. Only a small part of the chromium that ends up in water will eventually dissolve.
Crops contain systems that arrange the chromium-uptake to be low enough not to cause any harm. But when the amount of chromium in the soil rises, this can still lead to higher concentrations in crops. Acidification of soil can also influence chromium uptake by crops. Plants usually absorb only chromium (III). This may be the essential kind of chromium, but when concentrations exceed a certain value, negative effects can still occur.
Chromium (III) is an essential nutrient for humans and shortages may cause heart conditions, disruptions of metabolisms and diabetes. But the uptake of too much chromium (III) can cause health effects as well, for instance skin rashes. Chromium (VI) exposure may come from steel and textile industries as well as from smoking. The health hazards associated with exposure to chromium are dependent on its oxidation state. The hexavalent form, Cr(VI), is toxic and has adverse effects on the skin like ulcerations, dermatitis, and allergic skin reactions. Inhalation of Cr(VI) compounds can result in ulceration and perforation of the mucous membranes of the nasal septum, irritation of the pharynx and larynx, asthmatic bronchitis, bronchospasms and edema. Respiratory symptoms may include coughing and wheezing, shortness of breath, and nasal itch. According to National Toxicology Program, NTP, there is sufficient evidence for carcinogenicity in experimental animals for some hexavalent chromium compounds [18] .
For this reason, it is essential to remove lead and chromium from water and other environmental elements. Oswald, et al. [19] reviewed the methods of chromium removal and concluded that adsorption offered significant advantages like low cost, availability, profitability, ease of operation and efficiency, in comparison with conventional methods (such as membrane filtration or ion exchange) especially from economical and environmental points of view. Miretzky, et al. [20] used dead macrophytes for the removal of heavy metal ions like Cd2+, Ni2+, Cu2+, Zn2+ and Pb2+ from water. Lead was the most efficiently removed from the solution. Carmona, et al. [21] employed experimental design for the evaluation of the performance of biosorption of chromium for two levels of the operating parameters for a wide range of metal ion concentration. Barrado, et al. [22] attained optimal heavy metal removal (>99%) by precipitation as magnetic ferrites by treating wastewater with iron(II) sulfate for 2 hours at 50˚C and pH 10 in the presence of in a ratio [Fe(II)]/[total metal] of 15. Gaikwad, et al. [23] employed factorial design analysis for the study of copper removal from acid mine drainage by ion exchange.
The focus of the present work will be on the utilization of a certified Iraqi clay mineral, montmorillonite for the removal of chromium and lead individually and simultaneously from water and the use of Experimental Design in the planning of experiments and the analysis of the results to achieve the most favorable conditions.
2. Materials and Methods
2.1. Apparatus
Lead and chromium concentrations were measured with induced coupled plasma optical emission spectrometry on ICP-MS Thermo X-Series II, Thermo-Electron, San Hose, CT).
2.2. Materials and Reagents
Stock solutions of lead and chromium (5000 mg/L were prepared from analytical grade reagents in 1% nitric acid. Nanopure water (Barnstead NanoPureII, Dubuque, IA) was used throughout the study. The clay mineral used was a high quality Montmorillonite claystone of Wadi Bashira certified clay obtained from the Iraqi Geological Survey and with the following characteristics: Monmorillonite (77.0%); Loss on Ignition = 11.0; Organic Matter = 0.47; CEC = 0.7659 eq/g.
Buffer solutions were prepared from Pipes which is the common name for piperazine-N, N’-bis (2-ethanesul- fonic acid). A stock solution of 10 mM concentration was prepared from which the working pH buffers were prepared by simple adjustment of the main buffer with drops of 0.1 M HCl and 0.1 M NaOH.
2.3. Adsorption Studies
In 15-mL centrifuge tubes, a weighed amount of montmorillonite was placed together with 0.1 mL of 1.0 M KCl and 0.4 mL of the metal stock solutions. The final dilution was made to 10 mL with the pre-prepared buffer solution. All the sample solutions were prepared in duplicate. The sampling tubes were placed on a rotary shaker and run at a speed of 250 rpm for the predetermined times at the lab temperature which ranged between 19˚C - 21˚C. After equilibration, the samples were centrifuged at 3000 rpm for 10 mins to aid solid separation. The solutions were filtered through 0.22 µm syringe filters into plastic vials and kept at 4˚C for metal analysis.
The same procedure was repeated for solutions containing both chromium (III) and lead at a starting concentration of 200 mg/L. Aliquots of the filtered solutions were diluted with 1% HNO3 solution and kept for analysis by ICP MS.
3. Design of Experiments
The central composite design [24] was used in this work to allow studying the effects of three main parameters at five levels: equilibration time, 10 - 60 minutes; pH, 2 - 7.5 and clay content: 5 - 20 g/L. Jump 8 program was also employed for the analysis of the adsorption data. Five levels of each parameter were used and the coded and real values of the variables are listed in Table 1.
4. Results and discussion
The heavy metal sorption capacity, Cs (mg/L or mmol/kg), was calculated by Equation (1):
 (1)
(1)
where, Ccont and Ceq (mg/L) are the control and equilibrium aqueous metal concentrations, respectively, V (L) is the volume of solution, and m (g) is the mass of the clay. However, the initial concentrations of the Pb and Cr were not considered for the estimation of the amount adsorbed to avoid any possible loss of metal ions by adsorption onto the walls of the container.
The percent of metals sorbed to clay is calculated according to Equation (2).
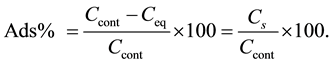 (2)
(2)
The Ads% and Cs values where then used in the statistical treatment of the results to evaluate the effects of operating parameters on the response.
4.1. Chromium Adsorption
The data of the chromium adsorption capacity were analyzed with the JUMP 8 program to predict the response
![]()
Table 1. Coded and real variable of the operating parameters used in the experiments.
values as a means for the assessment of the chosen model. The actual Cs values were correlated with the predicted values and the plot is shown in Figure 1. The correlation appears significant with a R2 value of 0.89 and a P value of 0.001 and RMSE value of 2.0311.
By ignoring the nonsignificant terms, the equation of predicting Cs values can be as follows:
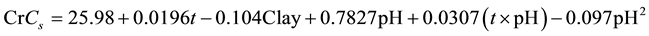 (3)
(3)
Figure 2 shows the effect of pH on the chromium adsorption capacity on 12.5 g montmorillonite/L at fixed Equilibrium Times. For a short equilibration time interval, 10 min, the maximum sorption capacity of not more than 16 mg/g was obtained at 12.5 g/L. The highest Cs could be obtained with 50 minutes equilibrium time for the same clay content. Further time interval does not help to improve the Cs further. Similarly, the Cr ? clay interaction was the highest, 21 mg/g, after 50 minutes of equilibrium at nearly neutral medium, pH = 6.39. The low pH medium is not favorable for chromium sorption on montmorillonite due to the expected desorption and high solubility of chromium species. Thus, chromium can be best adsorbed on montmorillonite at neutral medium for a clay content of 12.5 g/L of the clay. This is in agreement with the published reports [Carmona, et al, 2005] which revealed the impact of pH as the most significant effect regarding Cr3+ uptake on the biosorbent (Sargassum sp.). Also, Pandey, et al. [25] reported that the adsorption of Cr(VI) decrease with pH increase. Thus, the chemistry of Cr is the determining factor in the adsorption and not the sorbent properties. Wehrli, et al. [26] attributed the dependence of Cr adsorption on the pH to the fact that the protons are involved in the kinetic equation of Cr adsorption on aluminum hydroxide.
Maximum Cs, of chromium (25 mg/g) could be obtained with relatively low clay content of 8 g/L at a pH of 5. The sorption capacity is inversely proportional with the clay content. However, a clay content of 8 g/L gave identical Cs to the 5 g/L over a wide range of equilibrium time of 15 - 50 minutes. Longer contact time may not be recommended at slightly acidic solutions as a slight decrease of Cs could occur. Although low clay content may be in favor of increasing the quotient of adsorbed Cr on the clay content, the low clay content was not enough to give the required adsorption. It seems that there exists a compromise between clay content and the other conditions where the best Cr removal could be obtained with 8 g/L.
The Cs of chromium on montmorillonite is greatly affected by pH of the solution regardless of the clay content. The best performance could be noticed at a clay content of 12.5 at a nearly neutral pH (6.4). At low pH the long
![]()
Figure 1. Correlation of predicted chromium adsorption capa- city values with the actual values.
![]()
Figure 2. Effect of pH on the chromium adsorption capacity on 12.5 g/L of montmorillonite using various equilibrium times.
equilibrium time resulted in desorption of Cr from the clay surface. Meanwhile the long equilibrium improved the Cs for moderate acidic and neutral media. At a pH of 6.4, the Cs gradually increased with time between 16 and 22 mg/g within 40 minutes showing the effectiveness of the neutral medium in holding Cr on montmorillonite.
4.2. Chromium Adsorption Efficiency
Both pH and the clay content have significant impacts on adsorption efficiency of chromium on montmorillonite. After careful evaluation the following relation could be obtained for the Cr sorption efficiency:
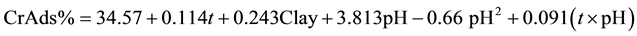 (4)
(4)
Regardless of the clay content and pH of the solution, equilibrium time of not less than 35 minutes is necessary to attain appreciable sorption. Figure 3(a) indicates that at least 13 g monmorillonite /L gave the highest Cr removal in slightly acidic medium (pH = 4.75). However, Figure 3(b) indicates that low pH values are not in favor of good sorption performance of Cr.
For the 250 mg/L concentration used, 60 minutes could only be adequate to cause 80% of the chromium removal on 12.5 g/L. With 16.7 g/L, only 40 minutes at a pH of 6.4 were enough to bring about total removal of chromium. Thus, a saturation value of 17 mg/g characterizes the process. At a pH value of 7.5, the saturation value increased to 17.85 mg/g after 40 minutes.
5. Adsorption Capacity of Lead on Montmorillonite
From the statistical treatment of the experimental results, it appears that all the operating parameters are affecting the adsorption capacity of lead on montmorillonite. However, the monmorillonite content is the most significant parameter in the process and the pH was the least significant. Excellent correlation exists between the predicted
adsorption capacity values and the real experimental response (R2 = 0.987). The lead Cs can be best decribed by equation (5):
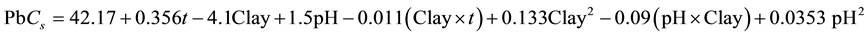 (5)
(5)
The response surface of the lead Cs is shown in Figure 4. The maximum Cs of lead (21 mg/g) occurs when the medium is nearly neutral (6.4) after 35 minutes. Figure 5 shows the effect of equilibration time on lead adsorp- tion capacity at pH = 4.75 and various clay contents. It is clear that even in slightly acidic medium lead is more efficiently sorbed on montmorillonite than chromium and increase with the increase of contact time specially with low clay content. The removal of lead was best done in acidic media: pH 3.0 using low cost bagasse [4] and pH 1.4 for lead removal from storage battery industry wastewater of an initial lead concentration of 4.2 mg/L using emulsion liquid membrane technique [4] .
Thus, the adsorption capacity of Iraqi montmorillonite for lead (24.28 mg/g, 0.1173 nM/g) is comparable to those reported for Saudi clay (30 mg/g, 0.147 mM/g) [5] and coconut shell carbon (26.50 mg g−1) [27] .
![]()
Figure 3. Effects of clay contents and pH on chromium adsorp- tion efficiency at various equilibrium times..
![]()
Figure 4. The response surface of lead adsorption capacity.
![]()
Figure 5. Effect of equilibration time on lead adsorption capacity at pH = 4.75 and various clay contents.
The effect of pH is not significant on lead Cs as we can reach considerable value at low pH by longer contact time. It was found that a clay content of 12.5 g/L gave 18.5 mg/g at a pH of 2.0 after 50 minutes and at pH of 7.5 after 20 minutes. With the neutral environment, pH = 7.5, the contact time have minor effect on the sorption capacity.
Lead Adsorption Efficiency
The overall relationship of the adsorption efficiency with the operatinng parameters can be expressed as in equation (6):
 (6)
(6)
Figure 6 shows the dependence of the sorption efficiency of Pb on montmorillonite on the clay content at a pH value of 4.75 and various equilibration intervals. The process is relatively slow and an equilibrium time of 10 minutes seems inadequate to give appreciable adsorption even with high clay content. Meanwhile, a clay content of 13 - 15 g/L gave maximum adsorption (94%) after 50 minutes. The adsorption efficiency undergoes slight in- crease with the increase of pH of the medium and the maximum can be attained beyond a pH of 6 after 40 minutes of equilibrium. Low clay content of 5 g/L appeared adequate to give appreciable Ads% (~90%) with long contact time (60 min.). The best case occurred with 16 - 17 g/L clay content, where a complete sorption of lead on mont- morillonite occurred after 50 minutes. Thus, the best operating conditions for the lead sorption are: pH = 6.4, time = 40 min. and clay content 16 - 17 g/L.
The response surface between pH and clay content after 35 minutes indicated a steady state of adsorption above a pH of 6.5 and clay content in the range of 10 - 15 g/L (Figure 7).
![]()
Figure 6. Effects of clay contents on lead adsorption efficiency at a pH of 4.75 and various equilibration times.
![]()
Figure 7. The response surface of lead adsorption efficiency for pH and clay content.
6. Simultaneous Adsorption of Pb and Cr by Montmorillonite
6.1. Sorption Capacity, Cs
When the two metal ions, Cr and Pb were present together, appreciable reduction in their concentration could be observed. Figure 8 and Figure 9 show the correlation of the actual Cs values with those predicted with the applied model for both Cr and Pb. For Cr, the correlation coefficient, R2 was 0.83, RMSE = 1.262 and p = 0.0069.
For Pb, the correlation coefficient, R2 was 0.92, RMSE = 1.1534 and p = 0.0002. Thus, the model fits better for lead than chromium and the montmorillonite particles can adsorb more lead ions than chromium. The clay content represents the denominator in the calculation of Cs and thus, its effect is negative on the adsorption capacity. The terms and their significance to the process are shown in Table 2 for Cr. The time and pH are the most significant parameters in the sorption of Cr on montmorillonite in addition to their combined action.
For Pb, the terms and their significance to the process are shown in Table 3. The statistical analysis indicated the significance of clay content and the pH on the lead adsorption. Also the combined actions of clay with pH and pH with time were found to be significant in the estimation of adsorption capacity.
![]()
Figure 8. The correlation of actual Cs Values with the predicted values for Cr in the presence of Pb.
![]()
Figure 9. The correlation of actual Cs values with the predicted values for Pb in the presence of Cr.
![]()
Table 2. Statistical analysis of the chromium adsorption data.
![]()
Table 3. Statistical analysis of the lead adsorptiondata.
The effect of clay content on the sorption capacity of chromium and lead in the presence of each other at a slightly acidic medium, pH = 4.75 and various contact times was evaluated. Regardless of the contact time the Cs of Pb decreased almost linearly with the clay content. For chromium moderate contact times (20 - 40 minutes) showed a maximum Cs with 10 - 13 g/L clay content. With long contact time, 60 min, the Cs of chromium showed a rapid drop with the increase of clay content. Clear improvement in the chromium Cs could be noticed with the increase of pH. However, at a clay content of 12.5g/L a steady level of Cs (10 mg/g) was obtained over the whole pH range employed.
The effect of contact time on the sorption Efficiency of chromium and lead on 12.5 g/L of montmorillonite at various pH values was also studied. The pH of the medium is a determining factor in a successful chromium sorption on montmorillonite. A slight increase of Cr Ads% with time and almost constant level, 71% - 75%, of Cr sorption could be obtained at nearly neutral medium, pH, 6.39, regardless of the contact time. Lead could be entirely removed from solution by sorption on 12.5 g/L of montmorillonite at a pH value of 6.39 after 60 minutes and after 35 minutes at a pH of 7.5. However, improved Cr adsortion, 86%, can be obtained at a pH value of 7.5 within 30 minutes. Thus, the most favorable conditions for simultaneous Cr and Pb may be 12.5 g/L clay contaent and a pH of 7.5 within 30 minutes.
6.2. Adsorption Isotherm
The plot of Log Cs against Log Ceq is given in Figure 10. The plot indicates a linear correlation over the clay content range of 5 - 17 g/L controls the adsorption of lead on montmorillonite with a correlation coefficient, R², of 0.9453:
![]() (7)
(7)
Cs values were used to calculate sorption coefficients as defined by
![]() (8)
(8)
where Kd (L/equiv) is the overall sorption coefficient, Ceq (mg/L) is the equilibrium aqueous Pb concentration, and CEC (meq/g) is the cation exchange capacity of the clay. The adsorbed Pb on solid was plotted against the equilibrium concentration in solution as in Figure 11. The overall adsorption coefficient was calculated by dividing the slop of the plot, 1.025 L/g, by the cation exchange capacity, CEC, of montmorillonite, 0.766 meq/g. A value of 1.338 L/meq was obtained for 250 mg/L Pb concentration level which is comparable with the value of the Kd values for sodium montmorillonite for relatively high iron concentration levels [28] .
7. Sorption Kinetics
Kinetics of metal sorption governs the rate, which determines the residence time, and it is one of the important characteristics defining the efficiency of an adsorbent [29] . Sorption kinetics can be controlled by several independent processes that could act in series or in parallel, such as bulk diffusion, external mass transfer (film diffusion), chemical reaction (chemisorption) and intra-particle diffusion [30] . The pseudo-second order kinetic
![]()
Figure 10. Adsorption isotherm of lead on montmorillonite..
![]()
Figure 11. Adsorption coefficient of lead on montmorillonite.
equation, developed by Ho et al., [30] :
![]() (9)
(9)
Or
![]() (10)
(10)
where k2 is the equilibrium rate constant for the Pseudo-second order adsorption (g・mg−1・min−1) and can be evaluated from the plot of t/Cs vs time. This model is based on the assumption that the rate limiting step may be a chemical adsorption involving valence forces through sharing or exchange of electrons between the adsorbent ans the adsorbate. Demirbas, et al., [31] applied this kinetic model successfully for the adsorption of metals.
Figure 12 shows the lead adsorption kinetic plots following pseudo second order model at pH values 3 - 7.5. Figure 13 shows the chromium (III) adsorption kinetic plots following pseudo second order model at pH values of 3 - 7.5. The plots indicated reasonable fit with the pseudo second order rate equation with correlation coefficient values, R2, values of 0.947 to 0.9967. The equilibrium adsorption capacity, Cs, was calculated from the slope of the plot and adsorption constant was calculated from the intercept of the linear plots. The results are presented in Table 4. From the table, the effect of pH on the equilibrium sorption capacity of chromium is clear. Meanwhile, the effect of pH on the lead equilibrium Cs is minor.
![]()
Figure 12. Lead adsorption kinetic plots following pseudo second order model at pH = ♦, 3; ■, 4.5; ▲, 6 and ●, 7.5.
![]()
Figure 13. Chromium (III) adsorption kinetic plots following pseudo second order model at pH = ♦, 3; ■, 4.5; ▲, 6 and ●, 7.5.
![]()
Table 4. Evaluation of the adsorption kinetics of chromium and lead on monmorillonite.
8. Conclusion
Experimental design was used to establish the most favorite conditions for the removal of chromium and lead from water by adsorption on a certified Iraqi montmorillonite. Effective adsorption capacity of 21.46 mg/g for chromium was obtained within 40 - 50 in a neutral medium. The acid medium resulted in low adsorption which suggested a desorption process for the adsorbed chromium. Almost 100% removal efficiency could be obtained for lead from water by adsorption on Iraqi montmorillonite under slightly acidic to neutral medium. The equilibrium could be attained within 45 - 50 minutes. The most favorable conditions for simultaneous Cr and Pb might be 12.5 g/L clay content and a pH of 7.5 within 30 minutes. The sorption results fit reasonably with the pseudo- second order kinetic model which also confirmed the pH dependence of chromium adsorption.
Acknowledgements
This work is supported by the Institute of International Education-Scholar Rescue Fund. The author is grateful to the dean and staff of college of Science and Mathematics, Montclair State University, NJ, USA for hosting him during the scholarship, to Dr. D. Sarkar, for his permission to use lab facilities to carry out this work; to Dr. H. Su for her help in the statistical treatment of lead results and to Dr. P. Punamiya for the help in the ICP measurements.
NOTES
*Partly presented at the Mid-Atlantic Regional Meeting of the American Chemical Society, MARM 2011, Maryland University, 21-24 May 2011.