Differential Deterrent Activity of Natural Products Isolated from Allophylus edulis (Sapindaceae) ()
1. Introduction
The use of synthetic pesticides in agricultural production yields effective results in the short term, but it has several drawbacks, such as the development of resistance and the adverse environmental effects. Among other approaches to substitute such products in Integrated Pest Management programs (pheromones, monitoring, and organic production), the developing of new pesticides from natural resources such as undamaged native plants (“botanical pesticides”) is becoming more and more comprised. These pesticides will be intrinsically less toxic and more environmentally friendly ways of control [1] .
In particular this study focuses on Allophylus edulis (Sapindaceae), commonly called Chal Chal, that occurrs in the Uruguayan and Brazilian native flora as well as in Bolivia, Argentina and the Guayanas (http://micol.fcien.edu.uy/flora/Allphyllus-edulis.html). Products obtained from A. edulis were evaluated against four pest species chosen to represent different feeding modes (chewing and sucking) and diet breadth (oligophagy and polyphagy). The four insect species, Spodoptera littoralis (Lepidoptera: Noctuidae), Epilachna paenulata (Coleoptera: Coccinelidae), Myzus persicae and Rophalosiphum padi (Hemiptera: Aphididae), are important agricultural pests, either in conventional or organic production [2] .
Various species belonging to the genus Allophylus have been studied on their biological activity. For instance, A. rubifolius and A. africanus revealed to act as antioxidants; the latter also exhibited antibacterial properties [3] [4] . Cytotoxicity is another property that has been described in species of this genus. In a study of A. timoriensis, its extracts showed to be cytotoxic to carcinogenic lung and colon cells [5] .
A. edulis has been scarcely studied. However, there are some reports about its antihepatotoxic activity [6] , its inhibitory action of the angiotensin converting enzyme [7] and its antiulcerogenic [8] and genotoxic activities [9] . The infusion of leaves is popularly used as throat anti-inflammatory agent and for intestinal disorders, as well as being considered antidiabetic [10] . As far as we know, anti-insect activity has only been reported by our group for the species A. edulis when tested against the aphid M. persicae and the coleopteran E. paenulata. In that occasion no fractionation neither isolation of pure compounds had been pursued [11] . Regarding isolated compounds from this species, there are reports on the presence of cyanolipids in seeds [12] [13] , of the methyl inositol quebrachitol in twigs [14] , of phenolic compounds (responsible for the inhibitory action of the angiotensin converting enzyme) in branches and leaves [7] , as well as of C-glycosil flavones (with antihepatotoxic activity) in leaves [6] .
In this study the phytochemical characterization of the ethanolic extract from twigs of A. edulis has been pursued following a bioguided fractionation procedure, resulting in the isolation of various active compounds against different insect models.
2. Materials and Methods
2.1. General
IR spectra were obtained in a IR Prestige-21 Shimadzu Spectrometer in NaCl.
The NMR spectra were recorded using an AMX-500 Spectrometer (400 and 500 MHz for 1H and 100, 125 and 150 MHz for 13C) in CDCl3 in the case of 1 to 4. In the case of 5 and 6 spectra were recorded using a Bruker DPXAdvance Spectrometer (400 MHz for 1H and 100 MHz for 13C) in CDCl3 as well.
In the cases of 1 to 3 mass spectrometric analyses were performed in a Micromass Autospec spectrometer with direct sample introduction and electronic impact ionization. Low resolution mass spectra were obtained in a Shimadzu 2010 Plus Gas Chromatograph coupled to a Shimadzu QP2010 Mass Spectrometer, with direct sample introduction and electronic impact ionization. In the cases of 4 and 6 analyses were performed using a Bruker micrOTOF-Q II mass spectrometer (Bruker Daltonics, Billerica, MA, USA), equipped with ESI. The instrument was operated at a capillary voltage of 4.5 kV with an end plate offset of −500 V, a dry temperature of 180˚C using N2 as dry gas at 4.0 L/min and a nebulizer pressure of 0.4 bar.
High Performance Liquid Chromatography was developed in a Shimadzu LC-20AT Prominence, equipped with a SPD-M20A Prominence diode array detector and Silica Phase columns. A Macherey Nagel Nucleosil-100 (7 mm, 250 × 16 mm) column was used, with DCM/MeOH as mobile phase, and a flow of 8 mL/min, in the case of 6; and a Beckman Ultrasphere Si-100 (5 mm, 250 × 10 mm) column, with hex/AcOEt as mobile phase, a flow of 8 mL/min, in the cases of 1-4. Flash and Liquid Chromatographies were developed in silica gel MN Kieselgel 60 M (0.04 - 0.063 mm/230 - 400 mesh). For flash columns a FMI Lab Pump, RP-SY and a IWAKI Air Pump, AP-115S were used. Fractions were monitored by thin layer chromatography developed in aluminium Silica gel 60 G/UV254 plates (Macherey-Nagel), and spots were visualized by heating after spraying with Oleum reagent [H2O:acetic:acid:sulfuric acid (16:80:4)] or Anisaldehide/H2SO4 (95 mL EtOH, 4 mL H2SO4, 1 mL anisaldehyde) [15] .
2.2. Extraction and Isolation
Vegetal material was collected in the countryside of Canelón Chico (Latitude: −34.4836111˚, Longitude: −56.3238889˚), Uruguay. Plant material was identified by Prof. Eduardo Alonso-Paz (Cátedra de Botánica, Facultad de Química) and a voucher specimen was deposited at the Herbarium of Facultad de Química, Montevideo, Uruguay [11] . Plant material was then dried (48 h, 40˚C) and ground with a IKA MF10 grinder. The extract was obtained by maceration in ethanol of 764 g of dried twigs, extracting the vegetal material three times for 24 h, yielding 5.1%. The extract (36 g) was fractionated by vacuum liquid chromatography (VLC) in silica gel (using a 13 cm height and 6 cm diameter silica column). Fractions were eluted with 2 - 3 dead volumes (200 mL) of mixtures of hex/AcOEt and AcOEt/MeOH, increasing the polarity over fractions. Fourteen fractions were obtained. Some of them were further separated in flash chromatography and preparative HPLC (see above) obtaining six isolated compounds (1-6).
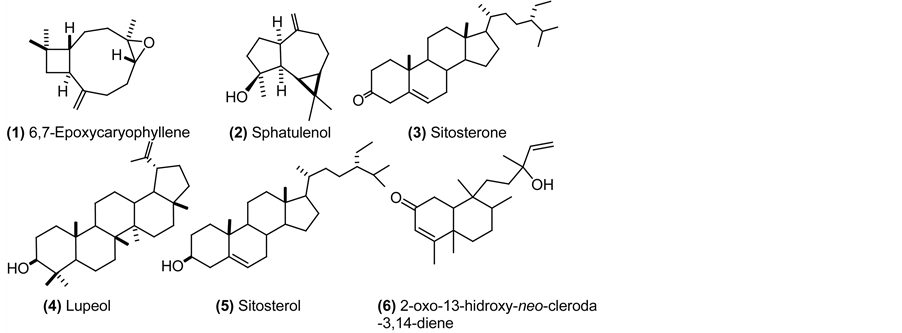
6,7-Epoxicaryophyllene (1): colorless oil (hexane); 1H NMR (CDCl3, 500 MHz) δ 0.96 (1H, m, H-8), 0.98 (3H, s, H-12), 1.01 (3H, s, H-13), 1.20 (3H, s, H-14), 1.32 (1H, m, H-5), 1.42 (1H, m, H-9), 1.65 (3H, m, H-1, H-9), 1.76 (1H, t, J = 10 Hz, H-10), 2.09 (2H, m, H-8, H-4), 2.25 (1H, m, H-5), 2.34 (1H, m, H-4), 2.60 (1H, dd J = 10, 18.5 Hz, H-2), 2.88 (1H, dd, J = 10.6, 4.3 Hz, H-6), 4.86 (1H, m, H-15), 4.97 (1H, d, J = 0,8 Hz, H-15). 13C NMR (CDCl3, 125 MHz) δ 17.2 (CH3, C-14), 21.7 (CH3, C-13), 27.4 (CH2, C-9), 30.0 (CH2, C5), 30.1 (CH3, C-12), 30.4 (CH2, C-4), 34.2 (C, C-11), 39.3 (CH2, C-8), 39.9 (CH2, C-1), 8.9 (CH, C-2), 51.0 (CH, C-10), 60.0 (C, C-7), 63.9 (CH, C-6), 112.9 (CH2, C-15), 152.0 (C, C-3). HREIMS m/z 220.1794 (calcd. for C15H24O, 220.18271).
Sphatulenol (2): colorless oil (hexane); 1H NMR (CDCl3, 500 MHz) δ 0.47 (1H, dd, J = 11.3, 9.5 Hz, H-1), 0.71 (1H, ddd, J = 11.3, 9.5, 6.1 Hz, H-2), 1.01 (1H, m, H-3), 1.04 (3H, s, H-13), 1.06 (3H, s, H-12), 1.28 (3H, s, H-15), 1.32 (1H, m, H-10), 1.57 (1H, m, H-8), 1.63 (1H, m, H-7), 1.77 (1H, m, H-8), 1.91 (1H, m, H-7), 1.98 (1H, m, H-3), 2.04 (1H, m, H-4), 2.20 (1H, m, H-6), 2.42 (1H, dd, J = 13.5, 7.0 Hz, H-4), 4.66 (1H, d, J = 1 Hz, H-14), 4.69 (1H, bs, H-14). 13C NMR (CDCl3, 125 MHz) δ 16.5 (CH3, C-13), 20.4 (C, C-11), 24.9 (CH2, C-3), 26.2 (CH3, C-15), 26.9 (CH2, C-7), 27.6 (CH, C-2), 28.8 (CH3, C-12), 30.1 (CH, C-1), 39.0 (CH2, C-4), 41.9 (CH2, C-8), 53.5 (CH, C-6), 54.5 (CH, C-10), 81.1 (C, C-9), 106.4 (CH2, C-14), 153.6 (C, C-5). HREIMS m/z 220,2002 (calcd. for C15H24O, 220, 18271).
Sitosterone (3): amorphous solid (hexane); 1H NMR (CDCl3, 500 MHz) δ 0.71 (3H, s, H-18), 0.83 (6H, m, H-26, H-27), 0.85 (3H, m, H-29), 0.92 (5H, m, H-21, H-9, H-24), 1.02 (2H, m, H-14, H-7), 1.12 (2H, m, H-15, H-17), 1.17 (4H, m, H-23, H-22, H12), 1.18 (3H, s, H-19), 1.25 (2H, m, H-28), 1.33 (2H, m, H-2, H-22), 1.36 (1H, m, H-20), 1.43 (1H, m, H-11), 1.51 (2H, m, H-11. H-8), 1.61 (1H, m, H-15), 1.67 (1H, m, H-25), 1.70 (1H, m, H-1), 1.84 (1H, m, H-6), 1.86 (2H, m, H-16), 2.02 (2H, m, H-1; H-12), 2.28 (1H, m, H-6), 2.39 (2H, m, H-2, H-7), 5.72 (1H, s, H-4). 13C NMR (CDCl3, 100 MHz) δ 12.13 (CH3, C-18), 12.15 (CH3, C-29), 17.6 (CH3, C-19), 18.8 (CH3, C-21), 19.2 (CH3, C-26), 20.0 (CH3, C-27), 21.2 (CH2, C-11), 23.3 (CH2, C-28), 24.4 (CH2, C-15), 26.3 (CH2, C-23), 28.4 (CH2, C-16), 29.4 (CH, C-25), 32.3 (CH, C-6), 33.1 (CH2, C-7), 34.1 (CH2, C-22), 34.2 (CH2, C-2), 35.8 (CH, C-8), 35.9 (CH2, C-1), 36.3 (CH, C-20), 38.8 (C, C-10), 39.8 (CH2, C-12), 42.6 (C, C-13), 46.1 (CH, C-24), 54.1 (CH, C-9), 56.1 (CH, C-14), 56.2 (CH, C-17), 123.9 (CH2, C-4), 171.8 (C, C-5), 199.8 (C, C-3). HREIMS m/z 412,3970 (calcd. for C29H48O 412.3705).
Lupeol (4): amorphous solid (hexane); 1H NMR (CDCl3, 500 MHz) δ 0.68 (1H, d, J = 9.2 Hz, H-5), 0.76 (3H, s, H-24), 0.79 (3H, s, H-28), 0.83 (3H, s, H-25), 0.94 (3H, s, H-27), 0.97 (3H, s, H-23), 1.03 (3H, s, H-26), 1.07 (2H, m, H-12), 1.28 (1H, m, H-9), 1.32 (1H, m, H-21), 1.36 (1H, m, H-18), 1.38 (1H, m, H-22), 1.39 (3H, m, H-6, H-7), 1.42 (2H, m, H-11), 1.48 (2H, m, H-16), 1.52 (1H, m, H-6), 1.57 (2H, m, H-2), 1.59 (2H, m, H-15), 1.66 (2H, m, H-1), 1.68 (4H, bs, H-13, H-30), 1.92 (1H, m, H-21), 2.01 (1H, m, H-22), 2.38 (1H, td, J = 11.1, 5.8 Hz, H-19), 3.19 (1H, dd, J = 11.4, 4.9 Hz, H-3), 4.57 (1H, dd, J = 2.5, 1.3 Hz, H-29), 4.68 (1H, d, J = 1.3 Hz, H-29). 13C NMR (CDCl3, 125 MHz) δ 14.7 (CH3, C-27), 15.6 (CH3, C-24), 16.1 (CH3, C-25), 16.3 (CH3, C-26), 18.2 (CH3, C-28), 18.5 (CH2, C-6), 19.5 (CH3, C-30), 21.1 (CH2, C-11), 25.2 (CH2, C-12), 27.58 (CH2, C-2), 27.6 (CH2, C-15), 28.2 (CH3, C-23), 29.9 (CH2, C-21), 34.5 (CH2, C-7), 35.8 (CH2, C-16), 37.3 (C, C-10), 38.7 (CH2, C-1), 38.2 (CH, C-13), 39.0 (C, C-4), 40.2 (CH2, C-22), 41.0 (C, C-8), 43.0 (C, C-14), 43.2 (C, C-17), 48.2 (CH, C-19), 48.5 (CH, C-18), 50.6 (CH, C-9), 55.5 (CH, C-5), 79.2 (CH, C-3), 109.5 (CH2, C-29); 151.1 (C, C-20). HRESIMS m/z 426.3821 [M]+ (calcd. for C30H50O: 426.38615).
Sitosterol (5): amorphous solid (hexane); 1H NMR (CDCl3, 400 MHz) δ 0.69 (3H, s, H-18), 0.80-0.85 (9H, m, H-27, H-29, H-26), 0.92 (5H, m, H-21, H-24, H-9), 1.01 (3H, s, H-19), 1.02 -1.04 (2H, m, H-17, H-22), 1.09 (1H, m, H-1), 1.12 (1H, m, H-14), 1.17 (3H, m, H-23, H-12), 1.25 (2H, m, H-28), 1.31 (1H, m, H-22), 1.36 (1H, m, H-20), 1.50 (5H, m, H-2, H-7, H-8, H-11), 1.59 (2H, m, H-15), 1.68 (1H, m, H-25), 1.85 (3H, m, H-1, H-2, H-16), 1.96 (1H, m, H-7), 2.02 (1H, m, H-12), 2.28 (2H, m, H-4), 3.51 (1H, m, H-3), 5.3 (1H, m, H-6). 13C NMR (CDCl3, 100 MHz) δ 12.0 (CH3, C-29), 12.1 (CH3, C-18), 18.9 (CH3, C-21), 19.2 (CH3, C-26), 19.6 (CH3, C-27), 20.0 (CH3, C-19), 21.2 (CH2, C-11), 23.2 (CH2, C-28), 24.5 (CH2, C-15), 26.2 (CH2, C-23), 28.4 (CH2, C-16), 29.3 (CH, C-25), 31.8 (CH2, C-2), 32.0 (CH, C-8), 32.1 (CH2, C-7), 34.1 (CH2, C-22), 36.3 (CH, C-20), 36.6 (C, C-10), 37.4 (CH2, C-1), 39.9 (CH2, C-4), 42.4 (CH2, C-12), 42.5 (C, C-13), 46.0 (CH, C-24), 50.3 (CH, C-9), 56.2 (CH, C-17), 56.9 (CH, C-14), 72.0 (CH, C-3), 121.9 (CH, C-6), 140.9 (C, C-5). EIMS m/z 414 [M]+ 399, 381, 354, 329, 303, 273, 255, 231, 213.
2-oxo-13-hidroxy-neo-cleroda-3,14-diene (6): colorless oil (hexane); IR (NaCl) 3441, 2934, 2850, 1730, 1660, 1454, 1381, 1261, 1022, 902, 848, 802 cm-1; 1H NMR (CDCl3, 400 MHz) δ 0.8 (3H, s, H-20), 0.82 (3H, d, H-17), 1.10 (3H, s, H-19), 1.25 (3H, s, H-16), 1.29 (2H, m, H-11), 1.32 (2H, m, H-6), 1.48 (5H, m, H-7, H-12, H-8), 1.83 (1H, m, H-10), 1.87 (3H, d, J = 1.3 Hz, H-18), 2.33 (2H, m, H-1), 5.06 (1H, dd, J = 10.7, 1.2 Hz, H-15), 5.20 (1H, dd, J = 17.3, 1.2 Hz, H-15), 5.70 (1H, d, J = 1.3 Hz, H-3), 5.80 (1H, dd, J = 10.8, 17.6 Hz, H-14); 13C NMR (CDCl3, 100 MHz) δ 15.9 (CH3, C-17), 18.2 (CH3, C-20), 18.5 (CH3, C-19), 19.1 (CH3, C-18), 27.0 (CH2, C-7), 27.9 (CH3, C-16), 31.3 (CH2, C-11), 35.0 (CH2, C-6), 35.01 (CH2, C1), 35.7 (CH2, C-12), 36.0 (CH, C-8), 38.5 (C, C-9) 40.0 (C, C-5), 45.8 (CH, C-10), 73.3 (C, C-13), 112.2 (CH2, C-15), 125.7 (CH, C-3), 145.1 (CH, C-14), 172.0 (C, C-4), 200.0 (C, C-2). HRESIMS m/z 304.23995 (calcd. for C20H32O2: 304.24022).
2.3. Insects
Epilachna paenulata Germar (Coleoptera: Coccinellidae) is an olyphagous insect specialized in Cucurbitaceae. Adults were maintained on squash (Cucurbita pepo L.) under controlled conditions of temperature (20˚C ± 2˚C) and photophase (14L:10D).
Spodoptera littoralis Boisduval (Lepidoptera: Noctuidae) is a generalist species. The laboratory colony was kept on an artificial diet [16] at 25˚C ± 1˚C, >70% relative humidity, with a photoperiod of (16L:8D) in a growth chamber.
Rhopalosiphum padi L. (Hemiptera: Aphididae) is a grass specialist species. These aphids were reared on Hordeum vulgare L. foliage and maintained at 20˚C ± 1˚C, >70% relative humidity, and with a photoperiod of (16L:8D) in a growth chamber.
Myzus persicae Sulzer (Hemiptera: Aphididae) is a generalist species. These aphids were reared on bell pepper foliage (Capsicum annuum L.) at 25˚C ± 1˚C, >70% relative humidity, with a photoperiod of (16L:8D) in a growth chamber.
2.4. Aphid Settling Bioassay
The activity on aphid settling was tested in choice experiments as described elsewhere [16] . The extracts were tested in plastic boxes (3 × 3 × 1.5 cm) lined at the bottom with 2% agar (20 replicates per extract). Two leaf pieces (ca. 1 cm2) cut from the appropriate host plant (C. annuum and H. vulgare for M. persicae and R. padi, respectively) were placed on the agar and treated either with the extract at 100 μg/cm2 (in the case of pure compounds a 50 μg/cm2 dose was used) or the same amount of solvent (MeOH). Ten aphids were placed in the box and the percentage of aphids settled on each surface was recorded after 24 hours of exposure. A Preference Index was also calculated for each replicate as PI = [(%C − %T)/(%C + %T)], where %T and %C are the percentages of aphids settled on the treated and control leaf pieces, respectively. In this manner, a PI < 0 indicates settling stimulation and a PI > 0 indicates settling deterrence.
2.5. Feeding Deterrence Bioassay
The antifeedant (phagodeterrent) activity was evaluated in choice-bioassays in Petri dishes (9 cm × 1 cm) lined at the bottom with a layer of agar (2%). Insects were offered four leaf discs (1 cm2) of the appropriate host plant (C. pepo for E. paenulata and C. annuum for S. littoralis). Two of the discs (T) were coated with 100 μg of the extract or 50 μg of pure compounds (10 μL of a 10% or 5% MeOH solution respectively), and the other two (C) were treated with 10 μL of MeOH. Insects were tested individually as adults for E. paenulata (10 - 15 replicates per extract) and as larvae for S. littoralis (5 - 6 replicates per extract). To measure deterrent activity, a visual score of area consumed (0%, 12.5%, 25%, 37.5%, 50%, 62.5%, 75%, 87.5% or 100%) was assigned for all discs within the plate, and a preference index (PI) was determined for each replicate using the formula PI = (C - T)/(C + T), where C and T are the amounts consumed for the control and treatment of the leaves respectively [17] . In this manner, a PI < 0 indicates a feeding stimulant effect and a PI > 0 indicates an antifeedant (deterrent) effect.
2.6. Statistics
Bioassay data were analyzed by Wilcoxon Rank tests [18] . Antisettling activity determination was based on the number of aphids settled on the leaf treated with solvent (control) compared to the number settled on the leaf treated with the substance tested (treatment). Replicates with fewer than five aphids settled in total (on both leaves) were not considered. Antifeedant (deterrent) activity determination was based on the proportions of the consumption of the leaf treated with solvent (control) compared to the proportions of the consumption of the leaf treated with the substance tested (treatment). Replicates where no consumption was detected were not considered.
3. Results and Discussion
The ethanolic twigs extract of A. edulis had showed significant deterrence against M. persicae as well as against E. paenulata (data from our previous report [11] ). However, many of the fractions obtained from the VLC showed to be active also against R. padi, the other aphid tested in this study (Table 1). Indeed, there were more fractions active against aphids (13 out of 14) than against chewers (10 out of 14). On the other hand, as a general trend, when activity was found, fractionation caused a decrease in activity against aphids and an increased effect

Table 1. Anti-insect activity of fractions eluted from the Vacuum Liquid Chromatography (VLC) of the ethanolic crude extract from twigs of A. edulis. Results are shown as mean ± standard error (SE).
aPI = (C − T)/(C + T), where C and T are the amounts consumed for the control and treatment leaves respectively for the chewers insects; and PI = [(%C − %T)/(%C + %T)], where %T and %C are the percentages of aphids settled on the treated and control leaf pieces, respectively for the aphids used. bAnti insect activity for the twig extract was previously reported [11] . *and bold results denote significant difference between C and T (p < 0.05, Wilcoxon 2 tail rank test). The activity is deterrent when PI > 0, and stimulant when PI < 0). #Denotes significant difference between C and T (deterrent, p < 0.05, Wilcoxon 1 tail rank test). NT: not tested.
on chewers compared to the whole twig extract, with the extreme in fraction 11 in which an antifeedant effect against S. littoralis was found when no activity was detected in the crude extract from which fraction 11 was obtained. These observations may point to some kind of adding or even synergistic effect in the mixture when tested against aphids, and on the contrary, an antagonistic effect in the case of the chewers.
The first fraction eluted with hex/AcOEt (8:2), was deterrent against M. persicae and E. paenulata but settling stimulant on R. padi (PI = −0.4 ± 0.1, p < 0.05, Table 2). This settling stimulating effect was neither present in the crude extract nor recovered for the compounds isolated from this fraction (that is, compounds 1 - 4, Table 2 and Table 3); indeed compound 1 isolated from this fraction was deterrent against this species. Deterrent activity of 1 was also found against the other aphid, M. persicae and E. paenulata.
Compound 2 was also isolated from this same fraction. From the analysis of mono and bidimensional NMR spectra and comparing them with bibliography it was concluded that 1 corresponded to 6,7-Epoxicaryophyllene [19] and 2 to spathulenol (Spectra as well as a brief description of elucidation are included in the supplementary material) [20] . Being both of them sesquiterpenes usually found in plant species but reported here for the first time in A. edulis.
These compounds have shown anti insect activity against different species, according to previous reports. Spathulenol (2) which was not tested in this study owing to the insufficient quantity obtained, has been already reported as a component of active essential oils against A. aegyptii larvae [21] [22] ; and adults of Rhyzopertha

Table 2. Anti insect activity of pure compounds isolated by flash chromatography and HPLC from the VLC fractions. For comparison purposes the activity of the VLC fractions from which the isolated compounds were obtained are included before such compounds. Results are shown as mean ± standard error (SE).
aSee Table 1 for details on PI. *and bold results denote significant difference between C and T (deterrent, p < 0.05, Wilcoxon 2 tail rank test). SE: Standard error. NT: not tested.

Table 3. Compounds isolated from the different fractions eluted from the VLC.
dominica (Coleoptera: Bostrichidae), Tribolium castaneum (Coleoptera: Tenebrionidae) (repellence and fumigant toxicity) and Sitophilus zeamais (Coleoptera: Curculionidae) (contact and fumigant toxicity) [23] -[25] . In these cases the individual activity of such compound was not tested, however there is a study of repellence against Aedes aegypti and Anopheles stephensi (Diptera: Culicidae) in which the pure compound showed significative activity [26] . Regarding 6,7-Epoxicaryophyllene (1) which showed activity against both aphid species as well as against E. paenulata (Table 2), there are many previous reports of plant essential oils that contain it and have shown activity against different insects. Some examples include the larvicidal activity of essential oils against different mosquitoes species (A. aegypti [27] [28] , Culex pipiens [29] , A. stephensi, Anopheles culicifacies, A. albopictus, Culex quinquefasciatus [30] [31] , Anopheles anthropophagus) [32] ; the fumigant toxicity against Ephestia kuehniella and Ectomyelois ceratoniae (Lepidoptera: Pyralidae) [33] ; the feeding and oviposition deterrence against larvae and adults respectively of S. frugiperda [34] ; the contact toxicity against Drosophila melanogaster L. (Diptera: Drosophilidae) and S. zeamais [35] [36] ; and the fumigant toxicity against Sitophilus granarius [37] . In the case of the aphids here tested, similar activity against R. padi and M. persicae was found for this compound (Table 2). Previous reports describe the activity of this compound on the settling of R. padi [19] and M. persicae [16] . In the case of the chewers, 6,7-Epoxicaryophyllene (1) probed to be feeding deterrent against E. paenulata but not against S. littoralis (Table 2). 1 accounts for 0.7% of its original fraction (fraction 2). However, it showed almost the same activity than the fraction against M. persicae and E. paenulata, indicating that other active compounds are also present in Fraction 2. As far as we are concerned, no previous studies had reported the activity of this compound against E. paenulata or S. littoralis.
Compound 3, also isolated from fraction 2 (Table 2) showed in its mass spectra a molecular ion m/z 412 which suggested the molecular formula C29H48O. In the 13C NMR spectra, 29 signals were observed, confirming along with 1H NMR signals and by comparison with bibliography data, that 3 corresponded to sitosterone [38] , described in this study for the first time in this plant species. Sitosterone was tested in this study against M. persicae and S. littoralis, not being active against any of them. As far as we know, there are no reports of activity of this compound against insects in previous literature.
Compound 4, also isolated from fraction 2 (Table 2) showed in its mass spectra a molecular ion m/z 426 which suggested the molecular formula C30H50O. By comparing NMR data with bibliography it was concluded that 4 corresponded to the pentacyclic triterpene lupeol [39] isolated for the first time in this vegetal species and revealing activity against M. persicae and E. paenulata.
In the case of lupeol (4), our results show deterrent activity against M. persicae and E. paenulata. There is a previous study in which its inactivity against M. persicae and S. littoralis is reported [40] . The divergence in the result on M. persicae found with the present work may be due to different aphid instars used in the tests. Within the frame of phytochemical and biological activity studies of plant extracts, this compound has been isolated from many plant species [41] -[49] . Moreover it has been reported to be component of a fraction active against the leaf miners Ctenopsteutis obliquana (Lepidoptera: Tortricidae) in feeding deterrence bioassays. However, when tested as a pure compound, it did not show activity against those larvae [50] .
The consecutive fraction [second fraction eluted with hex/AcOEt (8:2)] showed activity against E. paenulata (PI = 0.8 ± 0.3, p < 0.05), being inactive against the other insect models. From this fraction pure 5 was isolated. It showed in its mass spectra a molecular ion m/z 414 consistent with the molecular formula C29H50O. In such spectra there were signals of the ions m/z 273 and 255, corresponding to the steroid tetracyclic fragment. The loss of the side chain originates the fragment m/z 273 which after the elimination of H2O generates the ion m/z 255. The NMR signals of 5, were very similar to those of 3 except for the presence of an hydroxyl moiety in C3 instead of a carbonyl. With bibliography data it was possible to confirm that 5 corresponded to sitosterol [51] [52] , which in this study revealed to be active against M. persicae and E. paenulata. Noteworthy is the fact that the oxidation state in C3 is the only difference between 3 and 5, which may indicate a correlation of the moiety in such position with the activity of these sterols against insects. Similar correlations have previously been reported involving the activity of phytosterols against insects [53] .
At least a hundred different sterols have been isolated from plants, being them of vital importance for their growing processes [53] . Sitosterol is probably the most abundant and common sterol in plants [54] -[57] . Furthermore, sterols play a critical role in all the organisms of the animal kingdom, owing to their basic role as components of cell membranes and in some cases as hormone precursors (including molting hormones). However, herbivorous insects do not have the capacity of synthesising cholesterol. That is why they depend on the ingestion of phytosterols which could be metabolized to cholesterol [54] . In spite of that fact, in many cases anti insect activity has been found on some plant sterols. In those cases of phytosterols that have their function in the basic metabolism of plants and present also defensive activity against insect, it would be interesting to reveal if plants that produce them do so in different concentrations, deposit them differentially in their organs and/or are able to increase the biosynthesis in response to damage (assuming dose-dependent effect). In this direction, our results show (Table 2) that isolated sterols exhibited in the case of 3 decreased activity and in the case of 5 increased activity compared to the fraction from where they were isolated.
The next fraction [eluted with hex/AcOEt (1:1), fraction 4 in Table 1] was active against both aphids and the coleopteran. After its fractionation, 6 was isolated from it. In its mass spectra a molecular ion m/z 304 was observed which suggested the molecular formula C20H32O2. The signals observed in the mass spectra, 1H and 13C NMR revealed that 6 is 2-oxo 13-hidroxy-neo-cleroda-3,14-diene, a diterpene belonging to the clerodane series [58] ; isolated for the first time in Stachys rosea (Labiatae) [59] , being the present study the first report of this compound in the Sapindaceae family. However, some clerodanes have recently been reported on this family, particularly in the species Dodonaea viscosa [60] [61] . Owing to the little quantity obtained of this compound, no bioassays were developed assessing its activity. Concerning the activity of this compound against insects, to our knowledge, there is in literature only one report of its activity against E. paenulata [62] . Moreover, the group of the clerodanes has been described mainly from Lamiaceae and Asteraceae, and they have exhibited a wide range of anti-insect properties as it has been reviewed previously [63] -[65] . Seeing that this compound was isolated in the present study from a fraction (fraction 4; Table 2) active against E. paenulata and M. persicae, it could be suggested that 6 is at least in part, responsible of such activity.
4. Conclusions
In summary, A. edulis is here reported to produce various natural products with anti insect activity which seems to present in some cases synergic effects and in other antagonic effects. Besides, these products exhibited differential activity against the insects tested, a desirable attribute when developing botanical pesticides.
Further studies on the optimization of the extraction of bioactive compounds from this vegetal species should be developed, in order to complete the description of the anti insect activity of natural products from this plant.
Acknowledgements
The authors would like to acknowledge Professor E. Alonso Paz for the identification of the vegetal material and Dr. Gabriela M. Cabrera (Departamento de Química Orgánica, UMyMFOR-CONICET, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Argentina) for the acquisition of the mass spectra. The awards of postgraduate scholarships from LATU-FQ and ANII are specially acknowledged by the author M. D. The work developed in Spain was supported by PEDECIBA, CSIC and OPCW.
NOTES
*Corresponding author.