Water Efflux in Red Blood Cells of Sickle Cell Patients under Spontaneous Deoxygenation ()
1. Introduction
After one century, SCD is still an important worldwide health problem without a final solution [1] . Its complexity, genetic origin [2] and the different molecular processes involved in its pathophysiology [3] have contributed to this situation. The presence of an abnormal hemoglobin (HbS) inside the RBC, causing a polymerization process under deoxygenation, is recognized as the molecular process contributing most to the pathogenesis of SCD [4] , and for this reason many efforts have been made in relation to its study [5] [6] [7] [8] [9] . Nevertheless, the deoxygenation also causes cell damage, dehydration and shrinking [10] .
Different membrane abnormalities have been documented in RBC from sickle cell patients [11] : loss of lipid bilayer asymmetry, altered rate of phosphatidylcholine flip- flop, abnormal spectrin-actin skeleton, as well as, defective association between ankyrin and spectrin in situ on sickle cell inside out vesicles. These cause functional abnormalities as an abnormal transport phenotype [11] [12] : increased influx of calcium, reduced efflux permeability to non-electrolytes as glycerol and ethylene glycol and net loss of monovalent cations. Specially, the net loss of monovalent cations has been directly related with direct cellular dehydration because of the cellular response to maintain the osmotic equilibrium [12] .
The water transport in RBC has been widely studied before [13] - [18] . Especially, the water efflux and influx through the membrane of RBC from sickle cell patients has been studied in oxygenated erythrocytes [19] and in cells under total deoxygenation [20] , however, no studies under spontaneous deoxygenation have been performed. On the other hand, after deoxygenation, no values of 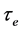 and no proper values of P have been reported.
and no proper values of P have been reported.
The deoxygenation pattern (deoxygenation % and speed) defines the HbS polymerization, as well as, the RBC shape change [2] [4] . In this work, we study 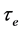 after HbS polymerization under spontaneous deoxygenation conditions. Different molecular mechanisms are considered to explain the observed behaviour, and the necessity of an adequate evaluation of the V/S to calculate P in sickle cells, after deoxygenation, is discussed.
after HbS polymerization under spontaneous deoxygenation conditions. Different molecular mechanisms are considered to explain the observed behaviour, and the necessity of an adequate evaluation of the V/S to calculate P in sickle cells, after deoxygenation, is discussed.
2. Materials and Methods
After the approval of the hospital ethical committee, and the obtaining of the informed consent, samples of RBCA (containing adult normal hemoglobin, HbA) and RBCS (containing HbS) were obtained [5] . Whole blood samples were obtained by venipuncture and immediately heparinized. After removing the plasma and leukocytes, RBC were washed three times with phosphate buffered saline (PBS, pH 7.4, Sigma Chemicals Co.) [5] [6] [7] [8] [9] . Supernatants were removed by centrifugation (500 g, 10 min) and decanting. The obtained washed RBC were resuspended (Hematocrit 45%, PBS (pH 7.4), 0.5% of BSA) and one portion was centrifuged (1000 g, 30 min, 25˚C) eliminating the supernatant to obtain packed RBC. 500 μl of packed RBC were taken and T2 for this sample was measured (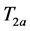 ).
).
300 μl of resuspended RBC were mixed with 600 μl of the manganese chloride stock solution (MnCl2(aq)) to obtain a final concentration of 2.5 mM. 500 μl of doped RBC were taken and T2 for this sample was measured (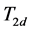 ).
).
Starting from the measured values of 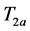 and
and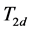 ,
, 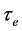 and P were calculated as follows [13] :
and P were calculated as follows [13] :
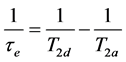 (1)
(1)
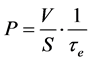 (2)
(2)
where V (0.63 × 10−10 cm3) and S (1.42 × 10−6 cm2) are the water volume inside the RBC and the surface of the RBC, respectively. V/S = 4.4 × 10−5 cm.
The magnetic resonance measurements were performed in a magnetic resonance console (MARAN DRX, Oxford Instruments, UK). T2 was measured using CPMG pulse sequence with a resonance frequency of 4.0353 MHz (homogeneous permanent magnet, B0 = 0.095 T), 10 and 20 µs for 90˚ and 180˚ pulses respectively, 128 scans, 50% for the RF power amplifier and the receiver gains, 3 s of relaxation delay and echo time of 0.2 ms (doped RBC) and 2 ms (packed RBC).
A student’s test (t-test) was performed to compare main values with α = 0.05.
3. Results and Discussion.
The values of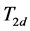 ,
, 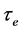 and P in oxygenated RBCA were measured for two temperature values (25˚C and 29˚C) (see Table 1).
and P in oxygenated RBCA were measured for two temperature values (25˚C and 29˚C) (see Table 1).
The 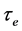 and P values for RBCA water efflux, shown in Table 1, are coherent with previous reports in the literature (
and P values for RBCA water efflux, shown in Table 1, are coherent with previous reports in the literature (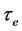 = (9.8 - 14.0) ms; P = (3.3 - 4.7) × 10−3 cms−1) [16] . The independence with the temperature is due to the activation energy characterizing
= (9.8 - 14.0) ms; P = (3.3 - 4.7) × 10−3 cms−1) [16] . The independence with the temperature is due to the activation energy characterizing
![]()
Table 1.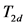 ,
, 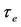 and P values corresponding to oxygenated RBCA for two temperature values (25˚C ± 1˚C and 29˚C ± 1˚C).
and P values corresponding to oxygenated RBCA for two temperature values (25˚C ± 1˚C and 29˚C ± 1˚C).
the process (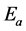 = 26 kJ∙mol−1) [18] and its influence in Equation (3).
= 26 kJ∙mol−1) [18] and its influence in Equation (3).
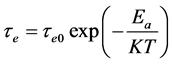 (3)
(3)
![]() , T and K are the initial water exchange time, the absolute temperature and the Boltzman constant, respectively.
, T and K are the initial water exchange time, the absolute temperature and the Boltzman constant, respectively.
The P values in oxygenated and spontaneously deoxygenated RBCA (24 h, 36˚C, 50% of deoxygenation) are shown in Figure 1. Because of the deoxygenation inhibits the KCC (K+ and Cl− cotransporter) channel activation in RBCA [12] , causing ![]() increases, these cells have a reduced P values after spontaneous deoxygenation. It was not taken into account and/or discussed in previous results [20] .
increases, these cells have a reduced P values after spontaneous deoxygenation. It was not taken into account and/or discussed in previous results [20] .
The values of![]() ,
, ![]() and P in oxygenated RBCS are summarized in Table 2. P
and P in oxygenated RBCS are summarized in Table 2. P
![]()
Figure 1. Efflux water permeability in oxygenated (100% of oxygenation) and spontaneously deoxygenated (50% of deoxygenation) RBC from healthy individuals. Red blood cells have intracellular concentrations and P was determined starting from ![]() and V/S ratio (Equation (2)).
and V/S ratio (Equation (2)). ![]() was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
![]()
Table 2.![]() ,
, ![]() and P values in oxygenated RBCS.
and P values in oxygenated RBCS.
decreases ((2.13 ± 0.26) × 10−3 cms−1) and ![]() increases (
increases (![]() = 20.89 ± 2.77 ms) if compared with the values obtained for healthy individuals (P = (3.27 ± 0.39) × 10−3 cms−1 and
= 20.89 ± 2.77 ms) if compared with the values obtained for healthy individuals (P = (3.27 ± 0.39) × 10−3 cms−1 and ![]() = 13.63 ± 1.67 ms) (see Figure 2).
= 13.63 ± 1.67 ms) (see Figure 2).
The increase in ![]() and P reduction in oxygenated RBCS, compared with healthy individuals, match with previous reports (P = (1.61 ± 0.39) × 10−3 cms−1 and
and P reduction in oxygenated RBCS, compared with healthy individuals, match with previous reports (P = (1.61 ± 0.39) × 10−3 cms−1 and ![]() = 29.2 ± 7.3 ms) [19] , and are closely related to:
= 29.2 ± 7.3 ms) [19] , and are closely related to:
・ The lipid peroxidation in the lipid bilayer.
・ The hemoglobin interaction with membrane Band 3 protein.
・ The oxidation of Band 3 sulfhydryl groups in the 15 kDa transmembrane fragment.
Because of simple diffusion only supports the 10% of the water transport through RBC membrane, the lipid peroxidation in the lipid bilayer cannot explain the observed P reduction in 34%. The sulfhydryl group oxidation is caused by the spontaneous generation of oxygen radicals and has been considered as the main contribution to P reduction [19] . The hemoglobin-membrane interaction is established with the Band 3 protein, at the same position as the main pore (4.5 Å) for water transport in RBC membrane; and, therefore, could cause an occlusion of this channel [20] . It has been demonstrated that this interaction is stronger in oxygenated sickle cell patient RBC than in cells from healthy controls [21] . As a consequence, the occlusion could contribute to the observed P reduction in oxygenated RBCS.
The values of ![]() and
and ![]() in RBCS, under spontaneous deoxygenation, are summarized in Table 3.
in RBCS, under spontaneous deoxygenation, are summarized in Table 3.
The spontaneous deoxygenation decreases ![]() values (
values (![]() =15.2 ± 1.0 ms) of the RBCS compared with the results obtained before deoxygenation (
=15.2 ± 1.0 ms) of the RBCS compared with the results obtained before deoxygenation (![]() = 20.8 ± 2.7 ms)
= 20.8 ± 2.7 ms)
![]()
Figure 2. Efflux water permeability in oxygenated (100% of oxygenation) RBC from healthy individuals and sickle cell patients. Red blood cells have intracellular concentrations and P was determined starting from the ![]() and V/S ratio (Equation (2)).
and V/S ratio (Equation (2)). ![]() was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
(see Figure 3).
Even though it is possible to find reports of the water transport for deoxygenated RBCS (P = (1.58 ± 0.06) × 10−2 cms−1) [20] , it does not affect the novelty of our results because in these experiments:
・ The reported experimental values of efflux water permeability were 10 times higher than the established values for human RBC [16] .
・ Induced complete deoxygenation was employed to provoke the HbS polymerization (0.5 mM, sodium dithionite).
・ The ![]() values are not reported.
values are not reported.
The explanation of decreased![]() , as a consequence of the spontaneous deoxygenation, is related with the hemoglobin-membrane interaction and the activation of the Psickle,
, as a consequence of the spontaneous deoxygenation, is related with the hemoglobin-membrane interaction and the activation of the Psickle,
![]()
Table 3. Values of ![]() and
and ![]() in RBCS under spontaneous deoxygenation.
in RBCS under spontaneous deoxygenation.
![]()
Figure 3. Water exchange time in oxygenated (100% of oxygenation) and spontaneously deoxygenated (50% of deoxygenation) RBC from sickle cell patients. Red blood cells have intracellular concentrations and ![]() was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
was measured using the T2 values of doped (2.5 mM MnCl2(aq)) and not doped RBC samples (Equation (1)). T2 was measured using CPMG.
Gardos, and KCC channels. The hemoglobin-membrane interaction could cause the appearance of pores starting from high order structure of the Band 3 protein [20] . We have previously demonstrated the increase of hemoglobin-membrane interaction with HbS polymerization under spontaneous deoxygenation conditions [22] . Now, we are strongly suggesting this increase could provoke the opening of new pores for the water efflux, which could explain the ![]() decrease after the agglutination process.
decrease after the agglutination process.
In RBCS, decreased oxygen partial pressure (PO2) provokes the Psickle activation, causing a K+ efflux, as well as, Na+ and Ca2+ influx. When the Ca2+ concentration reaches a threshold, the Gardos Channels (about 150 per cells) are activated causing a fast K+ and Cl− efflux [12] . As a difference with RBCA, in the RBCS the deoxygenation inhibits the KCC channel until PO2 = 40 mmHg, provoking it increases again when PO2 < 40 mmHg [12] . After 24 h of spontaneous deoxygenation (PO2 < 40 mmHg [23] , 50% of deoxygenation) KCC is reactivated in our experiments contributing to the net ions loss. The efflux of ions is coupled with a water efflux to maintain the osmotic equilibrium. The cellular dehydration increases the HbS concentration, which facilitates the HbS polymerization, the increase in the hemoglobin-membrane interaction, and the RBC shrinking.
On the other hand, it is important to consider that we are determining P (Equation (2)) for RBCA and RBCS using the same V/S ratio, as usual in the literature [19] . We consider this is an adequate procedure in oxygenated RBCS taking into account that: the Mean Corpuscular Volume (MCV) and Surface Area (S) do not have a statistically significant variation in these cells [24] - [29] . For deoxygenated RBCS is reported a MCV decreasing [30] [31] [32] [33] [34] and S remains constant [31] , which could change the V/S ratio and affect the P values. For this reason, in this work we are only reporting the ![]() values after deoxygenation for RBCS. To evaluate P, it is necessary to determine the V/S behavior in RBCS after deoxygenation. It could be done by performing a magnetic resonance experiment which includes a magnetic field gradient, studying the water self- diffusion coefficient behavior for short interpulse delay time in CPMG or stimulated echo pulse sequences [35] . Another way to measure P is starting from the effective water self-diffusion coefficient measured in the RBC, as well as, in the extracellular and intracellular medium [35] . Starting from this last determination and measuring, in the same sample, the
values after deoxygenation for RBCS. To evaluate P, it is necessary to determine the V/S behavior in RBCS after deoxygenation. It could be done by performing a magnetic resonance experiment which includes a magnetic field gradient, studying the water self- diffusion coefficient behavior for short interpulse delay time in CPMG or stimulated echo pulse sequences [35] . Another way to measure P is starting from the effective water self-diffusion coefficient measured in the RBC, as well as, in the extracellular and intracellular medium [35] . Starting from this last determination and measuring, in the same sample, the ![]() values using the method described in this work, it is also possible to determine V/S using Equation (2). It will be the subject of forthcoming works.
values using the method described in this work, it is also possible to determine V/S using Equation (2). It will be the subject of forthcoming works.
Not separated variations in water transport have been found in density-separated cell fractions of RBCS [19] [20] , for that reason our study has not considered it.
4. Conclusion
The HbS polymerization process, under spontaneous deoxygenation conditions, leads to an increase in the water transport through the RBC membrane from sickle cell patients, characterized by ![]() values of 15.2 ± 0.8 ms. The abnormal activation of the Psickle, Gardos, and KCC channels starting from deoxygenation, as well as, the possible appearance of new pores due to the increased hemoglobin-membrane interaction could explain this abnormal transport phenotype. The results obtained in this work increase the fundamental knowledge about molecular mechanism involved in SCD and could be useful in the development of new methods for diagnostic and treatment evaluation.
values of 15.2 ± 0.8 ms. The abnormal activation of the Psickle, Gardos, and KCC channels starting from deoxygenation, as well as, the possible appearance of new pores due to the increased hemoglobin-membrane interaction could explain this abnormal transport phenotype. The results obtained in this work increase the fundamental knowledge about molecular mechanism involved in SCD and could be useful in the development of new methods for diagnostic and treatment evaluation.
Acknowledgements
This work has been supported by the Belgian Development Cooperation through VLIR- UOS (Flemish Interuniversity Council-University Cooperation for Development) in the context of the Institutional University Cooperation program with Universidad de Oriente. The authors also want to thank the MRI RESEARCH CENTRE of the University of New Brunswick, Fredericton, Canada, for all the support received to finalize this work.