Subject Areas: Nuclear Physics, Particle Physics, Theoretical Physics

1. Introduction
BCS theory was proposed by John Bardeen, Leon Cooper, and John Robert Schrieffer (BCS) in 1957; they received the Nobel Prize in Physics for this theory in 1972. As an important nuclear theory, BCS [1] - [9] has made many important predictions. According to BCS formalism, the BCS ground state can be represented as:

Neutron-Proton (np) pairing effects play an important role in BCS theory. This paper does not consider np pairing; it only considers the nuclear lattice.
Unified Field Theory (UFT) [10] - [21] predicted the structures [10] - [13] of the proton and the neutron. According to UFT, a nucleon has three axes and a few other lower energy components. Each component plays different roles in nuclear stability. Two of three axes of a nucleon have 98% of the energy and they give nuclei the ability to strongly interact with one another and line up nucleons as a lattice. The lower energy axis for the proton is charged and for a neutron, in a heavy nucleus, is neutral. The weak interactions between lower energy axes line up nucleons along the third direction [10] .
Due to wave resonance, there are only a few configurations available for the lower energy components as they reach harmonic state over time. The massive axes [10] - [14] , roaming waves, and residential waves are weakly/ strongly interacting at bonding points and impacting lattice feature sites.
When an interaction in the nucleus reaches resonance state, the wave formation can be expressed as simplified BCS equation:
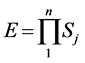 . (1)
. (1)
Si is an energy level in an electron mass unit or wave formation parameters. 2 * Si is an integer.
UFT consider the electron mass the quantum unit of particle/energy in nuclei as other energies resonant with charged energy. In the data book, the nuclear masses are in atomic mass unit. To convert it to an electronic mass unit:
1u = 1822.8884845e
It is difficult to get accurate atomic mass [22] . Using atomic mass in electronic mass unit is a novel technique deeply rooted in UFT similar to other concepts from UFT. Many recent physics papers [23] - [31] are studying similar issues.
This paper uses nuclear mass data and NMR results (Table 1) as experimental foundations.
1.1. UFT Concepts
The main structure of the Proton and Neutron are their axes [10] - [14] : A2, A2 and A (some time B).
The component “A” has the following mass formula from (1) in unit of electron mass:
A = 2 * 3 * 5 (charged)
In addition,
A2 = (2 * 3 * 5) * (2 * 3 * 5)
Component “B” has the following mass formula:
B = 2 * 2 * 4 (charged) or 2 * 2 * 2 * 2 (neutral)
![]()
Table 1. Masses for nuclei (experimental data).
1.2. Experimental and Theoretical Results
The experimental data and theoretical data (Table 2) in chapter 3 matched closely as follow:
The nuclear mass is based on the following formula:
Atom mass ?Z/1822.8884845 −(2 * Z + 8 * Z/4 + ∙∙∙)/(137 * 137 * 1822.8884845)
9Be and 25Mg can be calculated with a small margin of error. In the other case, the calculation is more precise. Noticeably, a neutron is at the center for 9Be and 25Mg in predicted lattices and bonding energy is smaller, while the calculation formula is complex. 10B also has a larger margin of error due to its special lattice introduced unexplained strong interactions.
The precise calculation of 27Al indicates that np paring is not an important factor in this theory. Even the worst result for 25Mg above is better than the existing theories, such as Ab Initio calculation [32] , and SEMF [33] . The precision of the above results demonstrates the merits of the new theory.
1.3. Configuration of Proton
If we use the electron mass as a unit, the mass of the proton (Figure 1) is:
1836.15267(e) = 2A2 + A + 2 * 3 + 0.15267
The structure formula is:
![]()
Table 2. Comparing experimental data and theoretical data.
2A2 + A + 2 * 3
The additional mass 0.15267 is related to various interactions:
1. The structure 2A2 strong interaction.
2. Weak interaction between charge and eight faces;
3. Weak interaction between 2 * 3 and A.
1.4. Configuration of Neutron
Using the mass of electron as a unit mass, we derive the mass of a neutron to be:
1838.68365987 = 2A2 + A + 2 * 3 + 2.5 + 0.15267 + 0.030987
The structure formula is:
2A2 + A + 2 * 3 + 2.5
The additional mass 0.15267 can be interpreted the same as proton. Energy 0.030987 is result of the following interactions:
1. Strong interaction between 2.5 and two A2 structure;
2. Dissonant weak interaction between 2.5 and 2 * 3;
3. Weak interaction between 2.5 and 2 * 3 * 5;
2. Results
2.1. Coupling of Proton and Neutron
When a proton and a neutron form a nucleus (Figure 2):
1. One of axes A2 from proton bonds with an A2 axis from neutron;
2. Wave 2 * 3 becomes 2 * 2;
3. The wave 2.5 of neutron becomes 2.
2.2. Two Neutrons and One Proton
Since there are two neutrons, two positive waves of energy level 3 are shared among them (Figure 3). A proton only contains three axes.
2.3. Symmetrical Preferences
Two pairs of interacting particles are arranged in the following manner:
![]()
Figure 2. Coupling of proton and neutron.
The black squares (Figure 4) are neutrons. The gray squares are protons. In the above figure, protons P1 and P2 form a pair, N1 and N2 form a pair of neutrons. These types of pairings are pp/nn pairings. The pairings in single layered nuclei do not play important roles as many stabled nuclei have odd number of neutrons or protons.
5H always decays to 3H as follow according to experimental results (Figure 5).
The two additional neutrons in 5H create complex energy wave pattern. 3H has simpler preferred structure.
For the same reason, proton rich 5Be has following decays confirmed by experimental results (Figure 6).
2.4. Isotopes of Helium
Helium (Figure 7) has two stable isotopes 3He and 4He.
2.5. Isotopes of Lithium
Lithium (Figure 8) has two stable isotopes 6Li and 7Li.
2.6. Isotopes of Beryllium
Beryllium (Figure 9) has one stable isotope 9Be.
8Be (Figure 10) has lower energy, but it can be decayed to stable structures.
10Be is (Figure 11) pretty stable, but it still can decay.
2.7. Isotopes of Boron
Boron (Figure 12) has two stable isotopes 10B and 11B.
2.8. Isotopes of Carbon
Carbon (Figure 13) has two stable isotopes and one relative stable isotope 14C.
Only 12C and 13C are stable. 14C (Figure 14) is less stable since it has a better alternative lattice 14N.
2.9. Isotopes of Nitrogen
Nitrogen has two stable isotopes (Figure 15).
2.10. Isotopes of Oxygen
Oxygen has three stable isotopes (Figure 16).
2.11. Isotopes of Fluorine
Fluorine has only one stable isotope (Figure 17).
2.12. Isotopes of Neon
Neon has three stable isotopes (Figure 18).
2.13. Isotopes of Sodium
Sodium has only one stable isotope (Figure 19).
2.14. Isotopes of Magnesium
Magnesium has three stable isotopes (Figure 20).
2.15. Isotopes of Aluminium
Aluminium has only one stable isotope (Figure 21).
2.16. Isotopes of Silicon
Neon has three stable isotopes (Figure 22).
2.17. Isotopes of Phosphorus
Neon has only one stable isotope (Figure 23).
3. Methods
The paper uses trial and error to find the possible nuclear structure for any lower mass isotope by trying various layouts and choosing the best symmetrical layout. The selected layout has to be verified by mass calculation according to the wave interaction of the given lattice.
Even though strong interaction has a relatively long range compared to the size of the nucleus, the strong interaction of nucleons are through bonding points where the neighboring nucleons interact with each other via overlapping axes. The strong interaction energy is 137e. Since the interactions are through axis A2 plus some additional energy, the strong interaction unit becomes:
137/(900 + E0)
This interaction unit is distributed among the interaction pockets E, such as nucleon count and energy number. The bonding energy at the bonding point resonances is:
137/((900 + E0) * E)
From Formula (1):
![]()
The weak interaction’s unit is:
![]()
When waves resonate with one another, the weak interaction energy is:
![]()
When waves are dissonant with one another, the compensate wave interaction factor is (can be strong or weak interaction):
![]()
The weak interaction energy becomes:
![]()
The interaction energy represents the energy transporting speed per wave cycle. Since a higher energy wave has a higher frequency, it makes the transportation throughput at a bonding point constant.
There are many structural components in a nucleon. The energy level of each component in a nucleon exists mainly for the stability of the nucleus itself. In a nucleus, the components in the nucleons become similar. This can reduce the complexity of many body interactions in a nucleus.
Conversely, each nuclear configuration is unique. A generic formalism cannot replace the case-by-case study on each isotope. We calculate the mass of new isotopes in this chapter.
Strong interaction of the internal waves can be visually represented as a rope model (Figure 24).
A = 2 * 3 * 5
The above formula is not just an equation; the formula shows how energy is formed like a rope (Figure 24). The inner wave is 2 (fine fiber of the rope), middle is 3 (fine string) and outer is 5 (the rope). The inner wave plus the middle wave 2 + 3 = 5. The strong interaction multiplies and aggregates from the center to the outer part. Fibonacci series are physically represented as energy waves like A2, B2, A, and B, from central wave 2 to outer wave 5 (or 4 in case of B).
When wave 2 * 2 interacts with A2, it interacts with the central waves of A2. Wave 2 * 3 interacts with middle and outer part of wave A2.
The roles in the nucleon are similar to a transportation system. The roaming wave is a passenger, bonding points are intersections, residential waves are vehicles, strong axes 2A2 are highways.
The roaming wave decides the next direction at the point of bonding between nucleons where the strong interactions take place. It chooses a vehicle randomly to make a turn or move straight.
Since wave 2 * 2 interacts with the inner wave 2 of A2, it helps passing wave moves straight when crossing the interacting point of 2A2. Wave 2 * 3 interacts mainly with wave 5 of A2. It helps the passing wave make directional changes or keep straight when crossing the interaction point of 2A2. Wave 2 + 3 helps the passing wave make directional changes and coupling with neighboring wave 2 * 2.
In a nucleus other than the hydrogen nucleus, A (2 * 3 * 5) changes to B(2 * 2 * 4) to resonant with the other nuclei properly by improving resonance with the inner wave 2 of A2. The wave B can be charged 2 * 2 * 4 or neutral 2 * 2 * 2 * 2.
3.1. Configuration of Proton
If we use the electron as the unit mass, we can see that the mass of the proton is:
1836.15267(e) = 2A2 + A + 2 * 3 + 0.15267
The additional mass 0.15267 is result of interactions in nucleus.
The structure 2A2 strong interaction:
137/900 = 0.152222
The weak interaction with charge vectors vertical to eight faces of octahedron structure is:
8/(137 * 137) = 0.000426
Wave 2 * 3 weakly interacts with A (2 * 3 * 5). 5 is not direct energy and it has a factor of 1/2. The self-dissonance of the weak interaction wave of 2 * 3 * 5 is:
2.5/(137 * 137 * 2 * 3) = 0.000022
This number matches exactly to the known Proton interactive mass 0.15267:
0.152222 + 0.000426 + 0.000022 = 0.15267
3.2. Configuration of Neutron
Using electron mass as a unit mass, we can calculate the mass of a proton to be:
1838.68365987 = 2A2 + A + 2 * 3 + 2.5 + 0.15267 + 0.030987
The Neutron is a Proton plus 2.5 mass waves. The 2.5 wave has a strong interaction with two A2 structure with bonding energy of:
137/(900 * 2.5 * 2) = 0.030444
For dissonant weak interactions between 2.5 and 2 * 3, 2.5 needs a factor of 2 to become 5. 2 + 3 = 5, but it is not direct energy. It has factor of ½:
P = 2 * 2.5
Q = 1/2
P/Q = (2 * 2.5/(1/2)) = 10
(P/Q)/(137 * 137) = 0.0005328
Weak interaction between 2.5 and 2 * 3 * 5 is related to the opposite charge. 2.5 needs factor 2 and 3 to interact:
1/(137 * 137 * 2 * 3) = 0.00001
0.030444 + 0.0005328 + 0.00001 = 0.030987
The number matches exactly with the known Neutron mass.
3.3. Coupling of Proton and Neutron
Proton:
2A2 + A + 2 * 3 + 137/900 + 8/(137 * 137) + (5/2)/(137 * 137 * 2 * 3)
Neutron:
Proton + 137/(900 * 2.5 * 2) + 10/(137 * 137) + 1/(137 * 137 * 6)
When a proton and a neutron form a nucleus, one of the axes A from proton bonds with an A axis from neutron introduce a new bonding force of 137/900. Wave 2 * 3 becomes 2 * 2 to resonant with two nodes.
The wave 2.5 of neutron becomes 2 to resonant with the other 2 * 2 waves. The dissonant weak interaction of 2.5 with 2 * 3 no longer exists since 2 * 3 wave changed to 2 * 2.
Wave 2 * 2 weakly interacts with A (2 * 3 * 5). 5 is not direct energy and it has factor of 1/2. The self-dissonance weak interaction wave of 2 * 3 * 5 is:
5/(137 * 137 * 2 * 2 * 3) = 0.000022
A transformed proton and neutron has lower energy in the 2H nucleus.
Proton:
2A2 + A + 2 * 2 + 137/900 + 8/(137 * 137) + 137/(900 * 3 * 2) + 5/(137 * 137 * 2 * 2 * 3) = 1834.15267
Neutron:
2A2 + A + 2 * 2 + 2 + 137/900 + 8/(137 * 137) + 5/(137 * 137 * 2 * 2 * 3) + 1/(137 * 137 * 6) = 1836.17805
Strong interaction on the bonding point:
1 * 137/900
Total: 3670.48294
It matches exactly to the known value: 3670.48294 of 2H mass.
3.4. Two Neutrons and One Proton
Since there are two neutrons, two negative waves 3 are shared among them. Proton only contains three axes:
3 * (2A2 + A) + 2 * 3 = 5496
Two energy 3 waves strongly interact with three nucleons:
2 * 3 * 137/900 = 0.9133333
The 2.5 wave in neutron is missing; the neutron bonding remains, as there are one more neutrons than protons. The energy is reduced to one quarter of neutron bonding:
137/(900 * 5 * 2 * 2) = 0.0076111
The weak interaction on eight faces of octahedron structure:
8/(137 * 137) = 0.000426
The weak interaction of two wave 3:
2/(137 * 137) = 0.000107
The total mass: 5496.9215
It matches the experimental data of 3H mass: 5497.9215
3.5. Two Protons and One Neutron
Since there is single neutron, only has single wave 5:
2 * (2A2 + A) + (2A2 + A + 5) = 5495
A2 aggregates with A during interaction (A2 + A = 930) as charged protons dominate the nucleus. Three nucleons strongly interact via two bonding points. But each bonding point has half the energy since the nucleons can rotate.
2 * 137/(930 * 2) = 0.147312
Dissonance wave of 5:
5 * 137/930 = 0.736559
Additional interactions are between 930, 30 and 5:
137/(930 * 30 * 5) = 0.001
The total mass: 5495.8851 matches the experimental data.
3.6. Two Neutrons and Two Protons
Since there are two neutrons, two positive waves 3 are shared among them. The charged axis A changes to B (2 * 2 * 4) to interact with the central waves of 2A2. Wave 2 * 3 facilitates passing wave’s direction changes. Energy formula becomes:
4 * (2A2 + B + 2 * 3) + 2 * 3 = 7294
Four nucleons strongly interact via four bonding points. But each bonding point has half energy since the nucleons can rotate. A2 aggregates with B during interaction (A2 + B = 916) as charged protons dominate the nucleus:
4 * 137/((900 + 2 * 2 * 4) * 2) = 0.29912664
Weak interaction:
4/137 * 137 = 0.0002131173
The total mass: 7294.29933975 matches the experimental data: 7294.299
3.7. 6Lithium (Figure 25)
Wave 2 * 2 helps straighten movement and wave 2 * 3 helps with turning. Wave 2 + 3 as shape effects for tilted 2 * 3 shape (Figure 25):
6 * (2A2 + B + 2 * 2 + 2 * 3) + 2 + 3 = 10961
Three nucleons strongly interact via seven bonding points, while two of them have half energy. A2 aggregates with B during interaction (A2 + B = 916) as charged protons dominate the nucleus:
6 * 137/(916) = 0.89738
Weak interaction:
6/(137 * 137) = 0.00032
Matches the experimental data: 10961.8977
3.8. 9Beryllium
Two pairs of proton lower 2 * 3 wave to 2 * 2 from Li. Its symmetrical shape makes two 2 * 2 waves as shape (Figure 26) effects:
9 * (2A2 + B + 2 * 2 + 2 * 2) + 2 * 2 + 2 * 2 = 16424
Strong interaction:
9 * 137/(926 * 5) = 0.2663
Closely matches the experimental data: 16424.2504
3.9. 10Boron
Two pairs of proton lower 2 * 3 wave to 2 * 2 from Li. The tilted 2, 3 and 4 nucleon lines can explain wave 3 + 4 as shape (Figure 27) effects:
10 * (2A2 + B + 2 * 2 + 2 * 2) + 3 + 4 = 18247
Three nucleons strongly interact via thirteen bonding points each shared by four parties. A2 does not aggregate with B during interaction similar to Beryllium case:
13 * 137/(900 * 4) = 0.456722
Additional unexplained strong interactions will be ignored:
137/(900 * 3 * 5) = 0.010148
Closely matches the experimental data: 18247.466754
3.10. 12Carbon
Three pairs of protons without an internal proton (proton is not paired with external proton) reduce 2 * 2 + 2 * 2 to 2 * 3. Only two or one protons in diagonal lines, they prefer interactive wave of 2 * 2 as square shape (Figure 28) effects:
12 * (2A2 + B + 2 * 3) + 2 * 2 = 21868
The nucleons strongly interact via sixteen bonding points in the center with A2 and 2 * 2:
16 * 137/(900 * 2 * 2) = 0.60889
Dissonant wave bonding:
2 * 137/(900 * 2 * 3) = 0.05074
Matches the experimental data: 21868.66
3.11. 16Oxygen
The twelve outer nucleons have wave 2 + 3 while four corner nucleons (Figure 29) have wave 2 * 3:
4 * (2A2 + B + 2 * 3) + 12 * (2A2 + B + 2 + 3) + 2 * 2 = 29148
The nucleons strongly interact via 24 bonding points in the center. A2 aggregates with B during interaction. Twenty four bonding points interact with 2 * 2:
24 * 137/(900 * 2 * 2) = 0.7611111
Dissonance of 2 * 3 and (2 + 3):
(2 * 3) * 137/(900 * (2 + 3)) = 0.182666
Interaction of B and (2 + 3):
137/(900 * (2 + 3) * 16) = 0.0019
This matches the experimental data: 29148.9457
3.12. 27Aluminium
The existence of internal proton prefers wave 2 + 3. Shared wave 2 * 2 as effects for the shape (Figure 30):
27 * (2A2 + B + 2 + 3) + 2 * 2 = 29148
The nucleons strongly interact via 44 bonding points. A2 does not aggregate with B during interaction as more neutrons are involved:
44 * 137/(900 * 2 * 2 * (2 + 3)) = 0.33489
It matches the experimental data: 49171.33606
3.13. 28Silicon
The existence of internal proton in 28Silicon (Figure 31) prefers wave 2 + 3 for external protons and neutron pairs. The six internal neutrons and two internal protons prefer 2 * 2:
20 * (2A2 + B + 2 + 3) + 8 * (2A2 + B + 2 * 2) + 2 * 2 = 50984
The nucleons strongly interact via 42 bonding points. A2 does not aggregate with B during interaction, E = 2 * 2 * 2 instead of 2 * 2 * (2 + 3) since wave 2 * 2 resonant with shared wave 2 * 2:
42 * 137/(900 * 2 * 2 * 2) = 0.7992
(2 + 3) strong interaction:
2 * 137/(900 * 2 * 2 * (2 + 3)) = 0.0152
Matches the experimental data: 50984.8144
3.14. Summary
Omitting the axes structural components, A2 and B, the main structural components are (Table 3).
4. Discussion
The energy calculations are studied for the lower mass elements. As elements become heavier, the strong interaction results become simpler and less accurate as the paper ignores smaller factors mainly related to weak interactions.
The structures of the nuclei are pretty certain for each isotopes, as it is impossible to find better alternatives for Carbon (Figure 32) as follows:
It is pretty obvious that there is no way to make 11Carbon (Figure 33) symmetrical:
In general, the symmetrical nuclei provide better circling passes for the roaming energy wave. Inner nucleons in proton/neutron pair allows roaming wave goes straight and it prefers wave 2 * 2. The external nucleons prefer 2 * 3 or 2 + 3 as the roaming wave has to change directions. The heavy mass elements prefer 2 * 2 * 4 (B) charge axes. For neutron, the B axes are neutral (2 * 2 * 2 * 2).
4.1. Nuclear Magnetic Resonance Sensitivity
1H (Figure 34) is the nucleus most sensitive to NMR signal. Proton NMR produces narrow chemical shift with sharp signals. Fast acquisition of quantitative results (peak integrals in stoichiometric ratio) is possible due to short relaxation time. The 1H signal has been the sole diagnostic nucleus used for clinical magnetic resonance imaging.
3He (Figure 34), is very sensitive to NMR. There is a very low percentage in natural helium, and subsequently has to be purified from 4He.
19F (Figure 34), relatively commonly measured. Sensitive, yields sharp signals, has wide chemical shift.
The energy calculations are studied for the lower mass elements. As elements become heavier, the strong interaction results become simpler and less accurate as the paper ignores smaller factors mainly related to weak interactions.
Not NMR sensitive (Figure 35):
The non-metal nuclei with stretched out configuration are more sensitive to NMR. Following is predicted configuration of 29Si (Figure 36):
Alternatively (Figure 37), it can have the following configuration:
Knowing that 29Si is a lot NMR sensitive than 28Si, it has to be configured as predicted structure with the following structural formula:
![]()
Figure 36. 29Si predicted lattice confirmed by NMR.
![]()
Figure 37. Alternative lattice not NMR sensitive.
22 * (2A2 + B + 2 + 3) + 9 * (2A2 + B + 2 * 2) + 2 * 3
4.2. Mass Calculation Confirmation
Once the structure of a nucleus is decided, energy of the nucleus can be analyzed. The accurate calculation result from simplified BCS formalism can be used to confirm the predicted nuclear structure.
24Mg, 25Mg and 26Mg (Figure 38) have following mass calculation:
24Mg: 24 * (2A2 + B + 2 + 3) + 2 * 3
25Mg: 25 * (2A2 + B + 2 + 3) + 3 * 3 + 25 * 137/(900 * 10)
26Mg: 26 * (2A2 + B + 2 + 3) + 2 + 3
5. Conclusion
A better understanding on nuclear structure is essential for future nuclear research. Unfortunately, detailed nuclear configuration cannot be observed visually. Since a nucleus is composed of neutron and proton, the nucleus is considered to be a product of many body interactions. The existing many body theories may be at a dead end and there have been no major developments since then. A new Unified Field Theory created more than ten years ago predicted detailed configuration of nuclei recently. The predicted configurations of nuclei can make nuclear “clear” and it narrows down the possible variations of nuclear structures. The proposed trial and error method not only can study nuclear configuration for lower mass nuclei but also can study configuration of any isotope of known mass. We anticipate our essay to be a starting point for more sophisticated studies on nuclear physics. For example, structure for each isotope can be discovered with help of the methods demonstrated in the paper.