An Efficient Synthesis of 2,3-Diaminoacid Derivatives Using Phosphine Catalyst ()
1. Introduction
2,3-Diaminoacids and their derivatives have attracted a great deal of attention due to their application as key structural fragments of biologically active compounds [1] -[7] , and/or as ligands for metal complexes [8] -[13] . Various methods to prepare these compounds have been reported [1] [2] [4] [14] -[19] .
While studying the PPh3-catalyzed three-component coupling of ethyl propynoate, a nitrogen nucleophile, and an aldehyde [20] , we found an efficient synthesis of 2,3-diaminoacid derivatives from ethyl propynoate and phthalimide catalyzed by triphenylphosphine. Further details on this synthesis are described in this report.
2. Results and Discussion
Reaction of ethyl propynoate (1) with phthalimide (2) in the presence of a stoichiometric amount of triphenylphosphine at room temperature gave ethyl 2,3-diphthalimidoylpropanoate (3) together with ethyl 2-phthalimidoylpropenoate (4) (Scheme 1). The ratio of 3 and 4 depended on the solvent used (Table 1). 3 was produced when highly polar solvents, such as DMSO, DMF, acetonitrile, and ketone were used. In these cases, 3 was obtained at more than 70% with no formation of 4. When 2-pyrrolidone was used, a mixture of 3 and 4 was obtained in 44 and 19% yields, respectively. However, the dehydroamino acid derivative 4 was mainly produced when a less polar solvent was used [21] . In esters, ethers, halogenated hydrocarbons, and aromatic solvents, 4 was obtained in moderate to good yields with no formation of 3.
DMSO and 2-butanone were selected to examine the effect of reaction temperature on the reaction (Table 2). The reaction in DMSO at room temperature for a short reaction time gave mostly 4. However, when the reaction time was increased, the yield of 4 began to decrease, while the yield of product 3 began to increase. Compound 3 was obtained in 97% yield at 100˚C and 87% yield at room temperature (Table 2, entry 8 and 11). In the 2-butanone solvent, it was also found that higher temperatures gave a better yield of 3.
The concentration of the substrates did not affect the yield of 3; the reaction of 1 mmol of substrate 1, 2 mmol of phthalimide (2), and 1 mmol of PPh3 in 1, 2, or 5 mL of DMSO gave 3 in 81%, 87%, and 88% yields, respectively, which were not significantly different.
This reaction also proceeded under other phosphines (Table 3). For example, the reaction progressed efficiently, when triphenylphosphine, tri(p-fluorophenyl)phosphine, and tri(p-methoxyphenyl)phosphine were used. These phosphines were found to act as catalysts [22] . For example, the reaction at room temperature for 24 h using 10 mol% triphenylphosphine resulted in 82% yield of 3. In contrast, the yield significantly decreased when tributylphosphine was used. Moreover, no reaction occurred when pyridine was used [23] .
Other nucleophiles were also investigated. Trost et al. reported that 4 was formed in 95% yield from the reaction catalyzed by triphenylphosphine in a mixture of toluene and a buffer solution of acetic acid and sodium acetate at 105˚C [24] . They found that using p-toluenesulfoamide as a substrate gave a trace amount of 3. Di-
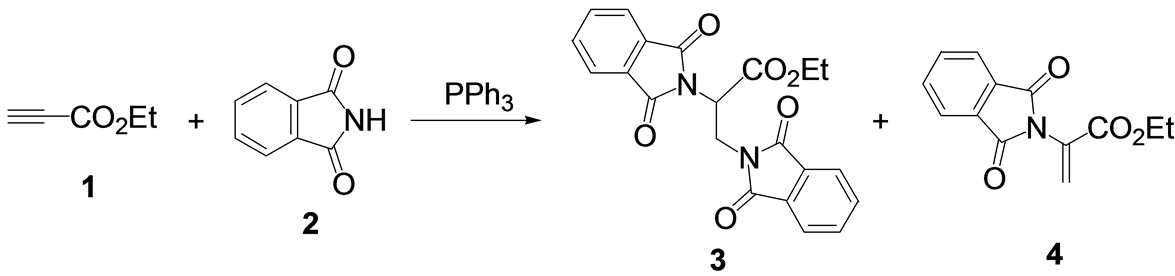
Scheme 1. Reaction of ethyl propynoate with phthalimide catalyzed by PPh3.
aA mixture of ethyl propynoate (1.0 mmol), phthalimide (2.0 mmol), PPh3 (1.0 mmol) was stirred in a solvent (2.0 mL) at room temperature for 24 h. bYields were determined by 1H NMR using internal standard (bibenzyl) method.

Table 2. Effect of reaction time and temperaturea.
aA mixture of ethyl propynoate (1.0 mmol), phthalimide (2.0 mmol), PPh3 (1.0 mmol) was stirred in a solvent (2.0 mL) at room temperature. bYields were determined by 1H NMR using internal standard (bibenzyl) method.
aA mixture of ethyl propynoate (1.0 mmol), phthalimide (2.0 mmol), phosphine (0.1 - 1.0 mmol) was stirred in dimethylsulfoxide (2.0 mL) at room temperature for 24 h. bYields were determined by 1H NMR using internal standard (bibenzyl) method.
acetamide, maleimide, benzylamine, allylamine, N-methyl-p-toluenesulfoamide, dimethyl malonate, and acetylacetone were also investigated, and they were found to give a 1:1 ratio of 3 and 4. Methyland phenylsubstituted propynoates were not converted to the desired 2,3-diaminoacid derivatives.
The reaction of 4 with phthalimide was also examined (Table 4). Without the presence of PPh3, 3 was not produced On the other hand, in the presence of PPh3, 3 was formed in good yield after 24 h. However, this reaction did not proceed in CH2Cl2.
The proposed reaction mechanism is shown in Scheme 2. First, triphenylphosphine attacks the propynoate to give zwitterionic intermediate 5 [25] -[39] . The intermediate subsequently remove a proton from phthalimide, giving a vinylphosphonium salt with a phthalimidate anion. The anion adds to the salt via Michael addition, followed by a proton transfer and elimination of phosphine to give 4. PPh3 attacks 4 again to give zwitterionic intermediate 7, which acts as a base to form another phthalimidate anion. This anion then attacks 4 by nucleophilic substitution to afford 3. Solvent effect is considered to stabilize zwitterionic intermediates 5 and 7.
3. Conclusion
α,β-Diamino acid derivatives are efficiently prepared by the reaction of propynoate with phthalimide in the

Table 4 . Synthesis 3 from the reaction of 4 and phthlimidea.
aA mixture of 4 (1.0 mmol), phthalimide (1.0 mmol), PPh3 (30 mol% or 0) was stirred in a solvent at room temperature for 24 h. bYields were determined by 1H NMR using internal standard (bibenzyl) method.
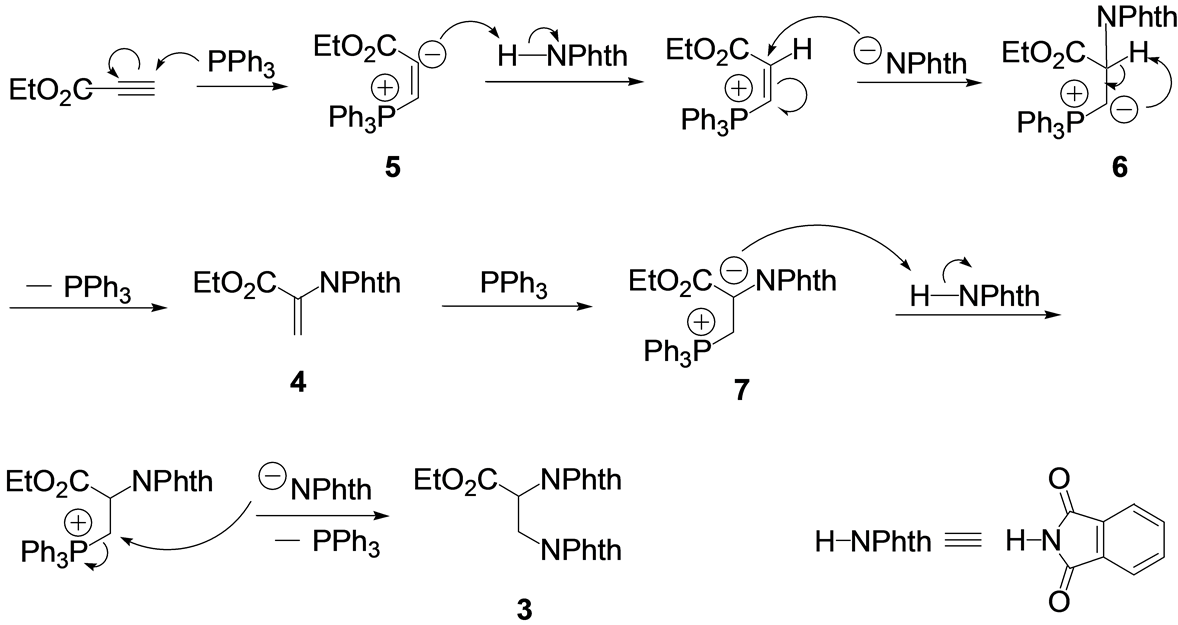
Scheme 2. Proposed mechanism.
presence of catalytic amount of triphenylphosphine. This method is very simple and the yield of the product is almost quantitative. Furthermore, if optically active amino acids could be prepared, this method might become more usable. Our first trial of the reaction using chiral phosphines gave racemic product. But now preliminary results using chiral additives indicate the possibility to yield the optically active product. Now establishment of asymmetric reaction is underway.
4. Experimental
4.1. General
Proton nuclear magnetic resonance (1H NMR) spectra were measured using a JEOL JNM A-400 (400 MHz) spectrometer using tetramethylsilane as the internal standard. IR spectra were measured on a Shimadzu IR-408 spectrometer. Mass spectral (GC-MS) data were recorded on a Shimadzu QP2000A instrument. Melting points were measured on a Yanako Model MP and were not corrected. All substrates were purchased and used without further purification except triphenylphosphine and phthalimide (recrystalization from methanol). Solvents were purified according to the literature method and stored under Ar atmosphere [40] .
4.2. Typical Experimental Procedure
Into a dry 80 mL Schlenk tube were added phthalimide (2.0 mmol), triphenylphosphine (0.3 mmol), and DMSO (2 mL). To this mixture, ethyl propynoate (1.0 mmol) was added dropwise, and the mixture was stirred for 24 h. The yield was determined by internal standard (bibenzyl) method. That is, bibenzyl (0.25 mmol) was added to the reaction mixture, and then concentrated mixture was analyzed by 1H NMR. The integration area of 5.17 ppm (product 3), 5.97 ppm (byproduct 4), and 2.91 ppm (bibenzyl) was used to determining the yields of 3 and/or 4. Reaction mixture was purified by column chromatography (silica gel 60, 200 - 400 mesh, hexane-ethyl acetate) to give the product.
4.3. Identification of the Products
Ethyl 2,3-diphthalimidoylpropanoate (3): Yellow solid, 1H NMR (CDCl3) δ 7.82 - 7.81 (m, 4H), 7.77 - 7.67 (m, 4H), 5.17 (dd, J = 9.2 , 5.6 Hz, 1H), 4.53 - 4.45 (m, 2H), 4.28 (q, J = 7.2 Hz, 2H), 1.27 (t, J = 7.2 Hz, 3H); 13C NMR (CDCl3) δ 167.8, 167.2, 166.8, 134.2, 134.1, 131.5, 62.3, 50.4, 36.8, 14.0; IR (KBr) 2960, 1760, 1724, 1600, 1380, 1220, 1085, 1030, 720, cm−1; GC-MS(m/z) 392; mp. 132˚C - 133˚C.
Ethyl 2-phthalimidoylpropenoate (4): White solid, 1H NMR (CDCl3) δ 7.91 - 7.87 (m, 2H), 7.79 - 7.75 (m, 2H), 6.66 (s, 1H), 5.97 (s, 1H), 4.26 (q, J = 7.14 Hz, 2H), 1.28 (t, J =7.12 Hz, 3H); 13C NMR (CDCl3) δ 166.5, 162.3, 134.5, 131.8, 129.4, 127.8, 123.9, 61.9, 13.9; IR (KBr) 3150, 2960, 1790, 1724, 1639, 1400, 1380, 1250, 1070, 1030, 720, cm−1; GC-MS(m/z) 245; mp. 71˚C - 72˚C.