Investigation of the serum oxidative stress in broilers fed on diets supplemented with nickel chloride ()
1. INTRODUCTION
Nickel (Ni) is the 5th most abundant element by weight after iron, oxygen, magnesium and silicon. And Ni is a naturally occurring element that can exist in various mineral forms and is used widely in the metallurgical, chemical and food processing industries, especially as catalysts and pigments [1]. The Ni salts of greatest commercial importance are nickel chloride (NiCl2), sulphate, nitrate, carbonate, hydroxide, acetate and oxide [1,2]. The essentiality of Ni in the nutrition of different classes of animals (including rats, pigs, goats and chicks and so on) has been demonstrated [3-6]. Ni is involved in methionine-folate metabolism [7-9], and is a composition of catalytically active hydrogenase protein [10] or might be part of a regulatory component [11]. Ni ions a higher affinity for proteins and amino acids and have shown to produce oxidation of proteins in cells [12]. Binding of Ni to some chromatin proteins in somatic cells may result in oxidative and structural damage to proteins [13]. It has been reported that Ni may have a role in the hormone action and in the regulation of the prolactin secretion [14,15]. Ni deprivation in rats can cause depressed growth, reduced reproductive rates, decreased sperm count and motility, and alterations of serum lipids and glucose [16], and also raise increased neonatal mortality, uneven hair development in pups, and result in ultrastructural changes and decreased cholesterol contents in the liver of the successive generations [4]. In addition, Ni is a nutritionally essential trace metal for micro-organisms and plants [17]. Currently, eight Nicontaining enzymes have been identified [18].
It has been reported that chemical substances (such as Ni) spread along a large area through wind, rain and so on, and are accumulated on plants, animals and soil, and can affect human health badly [19]. Higher quantity of Ni creates allergy, cancer, non malignant respiratory tract disorders and iatrogenic Ni poisoning, and may cause toxic effects in the immune system [20]. Also, Ni may bind to DNA-repair enzymes and generate oxygen-free radicals in various tissues in both human and animals, and enhance lipid peroxidation and finally cause protein degradation [21]. Free radical generation from the reaction of Ni-thiol complexes and molecular oxygen, and/or lipid hydroperoxides, could play an important role in the mechanism(s) of Ni toxicity [22].
However, there is no relevant research about the impact of NiCl2 on the oxidative stress in the serum of broilers. The purpose of this study was to investigate the effects of dietary NiCl2 on the oxidative stress and antioxidant function in the serum of broilers. Parameters used to represent the oxidative stress in this study included the activities of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GSH-Px), and the ability to inhibit hydroxy radical, and contents of glutathione hormone (GSH) and malondialdehyde (MDA).
2. MATERIALS AND METHODS
2.1. Chickens and Diets
240 one-day-old healthy avian broilers were randomly divided into four groups with 60 broilers in each group. Broilers were housed in cages with electrically heated units and were provided with water as well as undermentioned experimental diets ad libitum for 42 days.
A corn-soybean basal diet formulated by the National Research Council (NRC) [23] was the control diet. NiCl2·6H2O was mixed into the corn-soybean basal diet to produce experimental diets containing 300 mg/kg, 600 mg/kg and 900 mg/kg of NiCl2, respectively (Table 1). The references minimal and maximal concentration were used as Capcarova et al. introduced [24].
All experimental procedure involving animals were
Table 1. Composition of the basal diets for broilers (%).
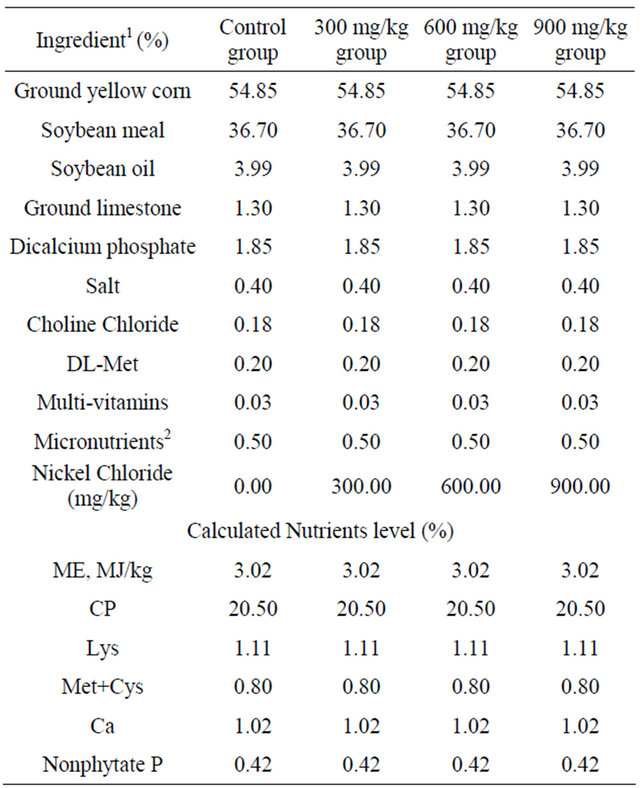
1Ingredient and nutrient composition are reported on as-fed basis. 2For the diet of d 1 - 42, provided per kilogram of diet: vitamin A (all-trans retinol acetate), 12,500 IU; cholecalciferol, 2500 IU; vitamin E (all-rac-α-tocopherol acetate), 18.75 IU; vitamin K (menadione Na bisulfate), 5.0 mg; thiamin (thiamin mononitrate), 2.5 mg; riboflavin, 7.5 mg; vitamin B6, 5.0 mg; vitamin B12, 0.0025 mg; pantothenate, 15 mg; niacin, 50 mg; folic acid, 1.25 mg; biotin, 0.12 mg; Cu (CuSO4 × 5H2O), 10 mg; Mn (MnSO4 × H2O), 100 mg; Zn (ZnSO4 × 7H2O), 100 mg; Fe (FeSO4 × 7H2O), 100 mg; I (KI), 0.4 mg; Se (Na2SeO3), 0.2 mg.
approved by Sichuan Agricultural University Animal Care and Use Committee.
2.2. Sample Preparation
5 broilers in each group were phlebotomized from the jugular vein at 14, 28, and 42 days of age during the experiment. Blood was clotted for 15 min and then centrifugated for 15 min at the speed of 3000 r/min. The serum was removed and assayed immediately.
2.3. Detection of Oxidative Stress Parameters in the Serum
The activities of SOD, CAT and GSH-Px, and ability to inhibit hydroxy radical, and contents of MDA and GSH in the serum were detected by biochemical methods following the instruction of the reagent kits (SOD: Cat.No.: A001-1, LOT: 20120625; CAT: Cat.No.: A007, LOT: 20120629; GSH-Px: Cat.No.: A005, LOT: 20120625; abilities to inhibit hydroxy radical: Cat.No.: A018, LOT: 20120624; MDA: Cat.No.: A003-1, LOT: 20120413; GSH: Cat.No.: A006, LOT: 20120629) which were purchased from Nanjing Jiancheng Bioengineering Institute of China. The absorbance of SOD, CAT, GSHPx, abilities to inhibit hydroxy radical, MDA and GSH were measured at 550, 240, 412, 550, 532 and 420 nm, respectively using a microtiter plate reader (Thermo, Varioskan Flash, USA).
2.4. Statistical Analysis
The significance of difference among four groups was analyzed by variance analysis, and results presented as means ± standard deviation (X ± S). The analysis was performed under SPSS 12.0 for windows. A value of p < 0.05 was considered significant.
3. RESULTS
3.1. Changes of the SOD Activities
The results were shown in Figure 1. The serum SOD activities were lower (p < 0.05) in the 600 mg/kg and 900 mg/kg groups than those in the control group at the 28 days of age, and were significantly lower (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups than those in the control group at the 42 days of age.
3.2. Changes of the CAT Activities
The serum CAT activities were reduced (p < 0.05) in the 900 mg/kg group at 28 days of age when compared with those of the control group, and were significantly lower (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups than those in the control group at 42 days of age, which was shown in Figure 2.

Figure 1. Change of the serum SOD activity (U/mL) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.

Figure 2. Change of the serum CAT activity (U/gHb) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
3.3. Changes of the GSH-Px Activities
As shown in Figure 3, the serum GSH-Px activities were lower (p < 0.05) in the 900 mg/kg group at 14 days of age, and significantly lower (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups than those in the control group from 28 to 42 days of age.
3.4. Change of the Ability to Inhibit Hydroxy Radical
The results in Figure 4 showed that the ability to inhibit hydroxy radical in the serum was reduced (p < 0.05) in the 600 mg/kg and 900 mg/kg group when compared with that of the control group at 14 days of age, and was significantly decreased (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups than that in the control group from 28 to 42 days of age.
3.5. Changes of the GSH Contents
The GSH contents were reduced (p < 0.05) in the 900 mg/kg group at 14 days of age, and were decreased (p < 0.05) in the 600 mg/kg and 900 mg/kg groups at 28 days of age, and were significantly reduced (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups at the 42 days of age when compared with those of control group. The results were shown in Figure 5.
3.6. Changes of the MDA Contents
The results in Figure 6 showed that the serum MDA

Figure 3. Change of the serum GSH-Px activity (U) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
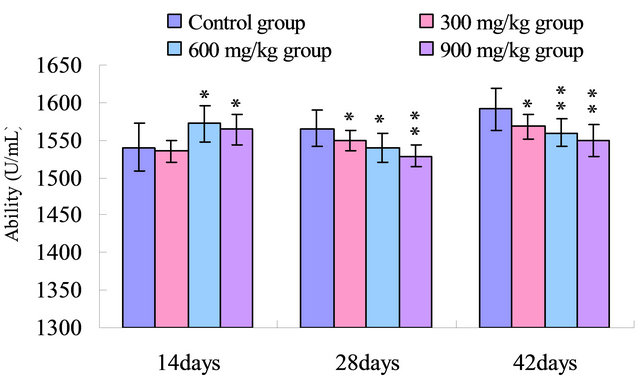
Figure 4. Change of the serum ability to inhibit hydroxy radical (U/mL) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.

Figure 5. Change of the serum GSH content (mg GSH/L) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.

Figure 6. Change of the serum MDA content (nmol/mL) in broilers. Note: Data are presented with the means ± standard deviation (n = 5); *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
contents were increased in the 900 mg/kg at 14 days of age, and were markedly increased (p < 0.05 or p < 0.01) in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups from 28 to 42 days of age when compared with those of control group.
4. DISCUSSION
Ni and other heavy metals can generate free radicals directly from molecular oxygen in a two step process to produce superoxide anion. A data suggests that antioxidants may play an important role in abating some hazards of Ni [25]. A number of studies have demonstrated that Ni can enhance lipid peroxidation in the liver, kidney, lung, bone marrow and serum of rats, and dose-effect relationships for lipid peroxidation in some organs have also been observed [26-31]. Lipid peroxidation may be a contributing factor in Ni-induced tissue oxidative stress [28]. NiCl2 also induces lipid peroxidation in rat renal cortical slices in vitro (ATSDR) [32]. It has been reported that a single intraperitoneal injection of nickel acetate increased lipid peroxidation and glutathione-S-transferase activity in the liver and kidney of rats [33]. In agreement with the abovementioned researches, our data suggested that NiCl2 induced the serum oxidative stress in broilers, which showed a dose and time dependent increase of MDA contents, decrease of GSH contents and GSH-Px, SOD, CAT activities, and inhibit hydroxy radical in the serum.
The cumulative production of reactive oxygen species/reactive nitrogen species ROS/RNS through either endogenous or exogenous insults is termed oxidative stress [21]. Endogenous antioxidants have the capability to prevent the uncontrolled formation of reactive oxygen negative ion. These antioxidants including CAT, GSH or mannitol provides the protection against the oxidative stress [34].
SOD and CAT, as the antioxidant enzymes, are considered to be the first line of cellular defense against oxidative damage [35]. In the present study, the activities of antioxidant enzymes including SOD, CAT and GSHPx in the serum were all decreased in the 300 mg/kg, 600 mg/kg and 900 mg/kg groups compared with those of the control group (Figures 1-3). The decreased activities of these enzymes can lead to an excessive availability of superoxide and hydrogen peroxide in biological systems, which in turn will generate hydroxyl radicals involved in the initiation and propagation of lipid peroxidation [36]. In our study, it was found that the ability to inhibit hydroxy radical was decreased in 300 mg/kg, 600 and 900 mg/kg groups when compared with that of the control group (Figure 4). Furthermore, hydroxy radical is one of the major oxygen radicals that can cause oxidative stress. Low level of antioxidants and high level of free radicals lead to the development of oxidative stress in the body.
Non-enzymatic antioxidants, such as GSH, play a primary role and regarded as an early biological marker of the oxidative stress [37]. In the present study, the GSH contents were significantly reduced in 300 mg/kg, 600 mg/kg and 900 mg/kg groups from 14 to 42 days of age (Figure 5). The reduced GSH was an important cellular antioxidant because of high intracellular concentration and also serves as a substratum of essential scavenger enzymes to maintain oxidative balance [38]. The decreased GSH-Px activity may also be due to the reduced availability of GSH in the present study.
As an end product of lipid peroxidation (LPO), MDA can cause cross-linking in lipids, proteins and nucleic acids [36,39,40]. And the MDA production induces alteration of membrane fluidity and increase of membrane fragility [41,42]. Moreover, MDA inhibits various enzyme reactions and exerts mutagenicity and carcinogenicity by forming DNA adducts [43]. Our results clearly showed increased MDA contents of the serum caused by dietary NiCl2 (Figure 6). As a late biomarker of oxidative stress, the increased production of MDA implies the enhancement of lipid peroxidation and accumulation of lipid peroxides in the body, which consequently reduces antioxidative function of the broilers.
According to the results observed in the present study and the aforementioned discussion, it is concluded that dietary NiCl2 in excess of 300 mg/kg can cause inhibition of antioxidant enzyme activities, enhancement of lipid peroxidation and accumulation of free radicals in the serum, which consequently induces oxidative stress and impairs the antioxidant function in broilers.
5. ACKNOWLEDGEMENTS
The study was supported by the program for Changjiang scholars and innovative research team in university (IRT 0848) and the Education Department and Scientific department of Sichuan Province (09ZZ017).