World Journal of Nano Science and Engineering Vol.05 No.01(2015),
Article ID:54964,6 pages
10.4236/wjnse.2015.51005
Controlling Diameter, Length and Characterization of ZnO Nanorods by Simple Hydrothermal Method for Solar Cells
Ahmed H. Kurda1, Yousif M. Hassan1, Naser M. Ahmed2
1Physics Department, College of Science, University of Salahaddin, Erbil, Kurdistan of Iraq
2Nano-Optoelectronic Research & Technology Laboratory, School of Physics, Universiti Sains Malaysia, Penang, Malaysia
Email: ahmedkurda.69@gmail.com, yousif.60@Hotmail.com, naser@usm.my
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 February 2015; accepted 20 March 2015; published 24 March 2015
ABSTRACT
Zinc oxide (ZnO) nanorods have been synthesized by solution processing hydrothermal method in low temperature using the spin coating technique. Zinc acetate dehydrate, Zinc nitrate hexahy- drate and hexamethylenetetramine were used as a starting material. The ZnO seed layer was first deposited by spin coated of ethanol zinc acetate dehydrate solution on a glass substrate. ZnO nanorods were grown on the ZnO seed layer from zinc nitrate hexahydrate and hexamethylene- tetramine solution, and their diameters, lengths were controlled by precursor concentration and development time. From UV-Visible spectrometry the optical band gap energy of ZnO nanorods was calculated to be 3.3 eV. The results of X-Ray Diffraction (XRD) showed the highly oriented nature of ZnO nanorods the hardest (002) peak reflects that c-axis elongated nanorods are oriented normal to the glass substrate. The Field Emission Scanning Electron Microscope (FESEM) was employed to measure both of average diameter of ZnO nanorods, Energy Dispersive X-Ray (EDX) is used to identify the elemental present and to determine the element composition in the samples.
Keywords:
Hydrothermal Method, Nanorods, Spin Coating, ZnO

1. Introduction
Zinc oxide (ZnO) is inexpensive n-type of semiconductor compound, which has shown promise for commercial applications in photovoltaic cells, [1] nanosensors, [2] [3] photocatalysise, [4] nanolasers [5] and light emitting diodes [6] . ZnO has a large band gap 3.37 eV, large excitonic binding energy 60 meV and high carrier mobility at room temperature. ZnO is composed of a hexagonal wurtzite crystal structure with unit cell a = 3.253 Å and c = 5.215 Å. Many reports have described syntheses ZnO nanorods through demonstrating various chemical processes that are simple and may also have industrial applications.
Nanostructured ZnO is fabricated using various thin film techniques as spray pyrolysis [7] , sputtering [8] , metal organic chemical vapor deposition [9] and hydrothermal method [10] . Hydrothermal method is widely adopted for the fabrication of transparent and conducting oxide due to its simplicity, safety, no needs costly vacuum system and hence cheap method for large area coating. The hydrothermal method also offers other advantages such as high surface area morphology at low crystallization temperature, the easy control of chemical components and fabrication of thin film at low cost for elucidating the structure and optical properties of ZnO nanorods.
In this work ZnO nanorods have been produced by thermal method using the solution, zinc acetate dehydrate in ethanol as a seed layer. The growth of ZnO nanorods, diameter and length are controlled by changing the solution concentration and immersion time in equimolar of zinc nitrate hexahydrate and hexamethylenetetramine (HMTA) in deionized water at a 90 ̊C and their morphologies, preferential orientation and optical properties were examined in particular.
2. Experimental Work
The hydrothermal method synthesis and thin film process arrangement are presented schematically in Figure 1 the glass substrate was cleaned with ethyl alcohol, urine, and acetone several times. The cleaned glass samples were further treated with UVO for 15 minutes to make rid of organic materials, the ZnO seed layer was first prepared as follows: A 5 mM ethanol solution of zinc acetate dehydrate (Zn(CH3COO)2∙2H2O, Aldrich, 98%)
Figure 1. The flow chart showing the procedure for preparation ZnO nanorods.
was spin coated on the cleaned glass at a spinning speed of 2000 rpm for 20 s with a 10 s wait time, then annealing at 150˚C for 15 min. The procedure was repeated three times, and finally the ZnO seed layer was annealed at 350˚C for 15 minutes. The solution of growing ZnO nanorods was prepared by dissolving equimolar zinc nitrate hexahydrate (Zn(NO3)∙6H2O, Aldrich 98%) and hexamethylenetetramine (HMTA) (C6H12N4, Aldrich, 99%) in deionized (DI) water. The solution concentration was varied from 15 to 35 mM for controlling the ZnO nanorods. The ZnO seed layer deposited on glass was immersed in the solution, where the glass was face down, and the baker was kept at 90˚C for 60, 90, 120, 150, 180 min. Alteration in the immersion time at a given concentration can control the length of the ZnO nanorods. The ZnO nanorods film was rinsed with Deionized water an ethyl alcohol several times. Ultimately, the film was annealed at 450˚C for 30 minutes.
The average diameter and length of the ZnO nanorods were measured by using the field emission scanning electron microscope FESEM (Model: FEI Nova NanoSEM 450). The transmission spectra of the films were measured by a double beam UV/visible (UV-4100) spectrophotometer with a wave length rang 200 nm - 800 nm and the optical band gap was measured from the transmission spectra.
X-Ray Diffraction (XRD) was utilized for the physical construction of the ZnO thin films. XRD patterns were obtained with a (Model: PANalytical X’pert PRO MRD PW 3040) single scan diffractometer with CuKα (λ = 1.54050 Å) radiation and scanning range of 2θ set between 20˚ and 80˚. The diameter and length of ZnO nanorods were measured using field emission scanning electron microscope FESEM (Model: FEI Nova NanoSEM 450). Energy Dispersive X-Ray Spectrometer (EDX) used for quantitative detection of elements in the prepared samples.
3. Results and Discussions
3.1. Optical Properties of ZnO Nanorods
Figure 2 shows the optical transmittance spectrum of nanocrystalline ZnO nanorods at 90˚C for precursor concentration 35 mM from immersion time 180 minutes annealed at 450˚C for 30 minutes using UV-Visible region from 200 nm - 800 nm. The transmittance is over 80% in the visible region from 400 nm to 800 nm for all the samples. Sharp absorption edge is located at 380 nm which is due to the fact that the ZnO is a direct ban gap semiconductor. The corresponding optical band gap of ZnO thin film is estimated by extrapolation of the linear relationship between ( hν)2 and hν according to Equation [11] .
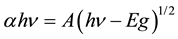 (1)
(1)
where α is the absorption coefficient, hν is the photon energy, Eg is the optical band gap and A is a constant. Figure 3 depicts the plot of (αhν)2 versus photon energy hν. The value of the direct optical band gap Eg is calculated from the intercept of (αhν)2 vs hν curve had also been plotted. The presence of a single slop in the plot suggests that the ZnO nanorod has direct and allowed transition. The band gap value of ZnO nanorod is found to be 3.3 eV which is slightly smaller to bulk ZnO (3.37 eV).
Figure 2. The transmittance spectrum of ZnO nanorods at 90˚C for pre- cursor concentration 35 mM from immersion time 180 min.
Figure 3. Plot of (αhν)2 vs photon energy hν of ZnO nanorods.
This difference is due to the fact the values of band gap Eg depend on many factors, e.g. the granular structure, the nature and concentration of precursors, the structural defects and the crystal structure of the films. Moreover, departures from stoichiometry form lattice defects and impurity stats. Dengue Bao et al. [12] reported that the band gap difference between the thin film and crystal is due to the grain boundaries and the imperfection of the polycrystalline thin films. D. L. Zhange et al. [13] reported that this band gap difference between the film and bulk ZnO is due to the grain boundary, the stress and the interaction potentials between defects and host materials in the films.
3.2. Structural Analysis of ZnO Nanorods
Figure 4 depicts the X-Ray Diffraction (XRD) pattern of the crystal structure and orientation of the nanocrystalline ZnO nanorods deposited on glass substrate using spin coating at 2000 rpm, pre-heated at 150˚C and annealed in air at 450˚C. From the XRD pattern, one can clearly observe a diffraction peak at 2θ = 34.426˚. Strong preferential growth is observed along c-axis, i.e. (002), suggesting that the prepared ZnO nanorods have the wurtizit structure.
The unit cell “a” and “c” of the crystalline ZnO nanorods with (002) orientation is calculated using the relation (2) and (3):
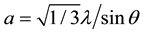 (2)
(2)
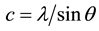 (3)
(3)
The values obtained for the unit cell a = 3.007 Å and c = 5.21 Å are in a good agreement with those reported in the JCPDS standard data (card no. 80 - 0074). The calculated parameters are given in Table 1.
From the XRD spectrum, grain size (D) of the film is calculated using debay scherrer formula [14] .
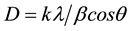 (4)
(4)
where k is a constant to be taken 0.49 [14] and, λ, β, and θ are the XRD wave length (λ = 1.5406 Å), full width at half maximum (FWHM) and Bragg angle respectively. By inserting the different values from Table 2 in the Scherrer formula grain size of (002) oriented thin film is 44.12 nm which is same as reported in literature [15] .
The dislocation density (δ), which represents the amount of defects in the crystal, is estimated from the following equation:
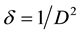 (5)
(5)
Strain ( ) of the thin film is determined from the following formula:
Figure 4. X-Ray Diffraction of the ZnO nanorods grown at 90˚C for 180 min from the 35 mM precursor concentration.
Table 1. Lattice parameters of the ZnO nanorods.
Table 2. Structure parameters of the ZnO nanorods.
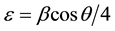 (6)
(6)
The calculated structural parameters of the thin film are presented in Table 2.
3.3. Morphological Analysis of ZnO Nanorods
In Figure 5, the (FESEM) shows the average diameter (d) of the ZnO nanorods increases from (57, 64, 83, 120 and 230 nm) as the precursor concentration increase from 15, 20, 25, 30, and 35 mM, respectively, where the immersion time is fixed for 180 min at 90˚C. Length of the grown ZnO nanorods is about 1 µm regardless of concentration, which indicates that changes in the precursor concentration at the fixed immersion time can affect only the diameter of the hexagonal ZnO nanorods. The rate of increase diameter of the ZnO nanorods is estimated to be approximately 34.4 nm/mM.
Length of ZnO nanorods can also be varied when the immersion time changes in the fixed concentration. Figure 6 shows that the average length of the ZnO nanorods increases from (241, 459, 522, 820 nm and 1.2 µm) as the immersion time t increases from 60, 90, 120, 150 and 180 min, respectively, at the precursor concentration of 35 mM. Length of ZnO nanorods indicates that growth rate is 6.3 nm/min.
Figure 7 shows the (EDX) spectrum and atomic composition of the ZnO/glass (002) layers for precursor con- centration 25 mM at immersion time 180 min. A description of the atomic composition of the elements in the layers is shown in percentages, as presented in the inset table in Figure 7, the concentration of these elements is indicated by the peaks, and clearly shows that the elements corresponding to the peaks comprised the layer. No contaminated element detected in the layers.
4. Conclusion
In this work, we have grown ZnO nanorods on glass substrates by solution processing hydrothermal method in low temperature using the spin coating technique. The structural, morphological and optical properties were in-


Figure 5. Surface FESEM images and diameter of hexagonal ZnO nanorods grown at 90˚C various concentrations (a) 15; (b) 20; (c) 25; (d) 30; and (e) 35 mM pre- cursor concentration for 180 min.


Figure 6. Surface FESEM images and length of hexagonal ZNO nanorods grown at 90˚C for immersion time (a) 60; (b) 90; (c) 120; (d) 150; and (e) 180 min from precursor concentration 35 mM.
Figure 7. The (EDX) of the ZnO nanorodes at 90˚C for precursor concentration 35 mM from immersion time 180 min.
vestigated. The hydrothermal method is a relatively simple technique: there are many factors which affected the quality of the film. We have optimized different parameters to obtain a good crystalline structure of ZnO nanorods with intense and sharp peak. The optical transmittance is over 80% in the wave length range from 400 nm - 800 nm and the band energy band gap is found to be 3.300 eV. According to XRD results, the as deposited films exhibited a hexagonal wurtized structure with (002) preferential orientation after annealing at 400˚C in air ambiance for 30 min. The XRD pattern consists of a single (002) peak which occurred due to ZnO crystals and grows along the c-axis. The grain size estimated to be 44.46 nm. The average diameter and average length of the ZnO nanorods obtained from the FESEM. The average diameter of ZnO nanorods, which are increasing from (57, 64, 83, 120 and 230 nm) as the precursor concentration increases at 90˚C for immersion time 180 min, and the average length of ZnO nanorods increases from (241, 459, 522, 820 nm and 1.2 µm) when the immersion time was increased at 90˚C for precursor concentration 35 mM. The (EDX) analyses of the samples clearly show that the sample prepared by above route has pure ZnO nanorod phases.
References
- Ku, C.H. and Wu, J.G. (2007) Electron Transport Properties in ZnO Nanowire Array/Nanoparticle Composite Dye- Sensitized Solar Cells. Applied Physics Letters, 91, Article ID: 093117.
- Cheng, X.L., Zhao, H., Huo, L.H., Gao, S. and Zhao, J.G. (2004) ZnO Nanoparticute Thin Film, Preparation, Charectrarazation and Gas-Sensing Properties. Sensors and Actuators B: Chemical, 102, 248-252. http://dx.doi.org/10.1016/j.snb.2004.04.080
- Kim, J.Y., Jeong, H. and Jang, D.J. (2001) Synthesis of a Graphene-Carbon Nanotube Composite and Its Electroche- mical Sensing of Hydrogen Peroxide. J. Nanoplast. Res., 13, 6699-6706.
- Chacrabarty, S. and Dutta, B.K. (2004) Photocatalytical Degradation of Model Textile Dyes in Wastewater Using ZnO as Semiconductor Catalyst. Journal of Hazardous Materials, 112, 269-278.
- Huang, M.H., Mao, S., Fieck, H., Yan, H., Wu, Y., Kind, H., Weber, E., Russo, R. and Yang, P. (2001) Room Temper- ature Ultraviolet Nanowirenano Lasers. Science, 292, 1897-1899.
- Saito, N., Haneda, H., Sekiguchi, T., Ohashi, N., Sakaguchiandk, I. and Koumoto, K. (2002) Low Temperature of Light- Emitting Zinc Oxide Micropatterns Using Self-Assembled Monolayer. Advanced Materials, 146, 418-421. http://dx.doi.org/10.1002/1521-4095(20020318)14:6<418::AID-ADMA418>3.0.CO;2-K
- Krunks, M. and Mellicov, E. (1995) Zinc Oxide Thin Films by the Spray Pyrolysis Method. Thin Soled Filme, 270, 33-36.
- Nunes, P., Costa, D., Fortunate, E. and Martiens, R. (2002) Performenes Presented by Zinc Oxide Thin Films Deposited by R. F. Magnetron Sputtering. Vacumm, 64, 293-297.
- Wu, J.-J. and Liu, S.-C. (2002) Low-Temperature Growth of Well-Aligned ZnO Nanarods by Chemical Vapor Deposition. Advanced Materials, 14, 215-218. http://dx.doi.org/10.1002/1521-4095(20020205)14:3<215::AID-ADMA215>3.0.CO;2-J
- Ni, Y.H., Wei, X.W., Hong, J.M. and Ye, Y. (2005) Hydrothermal Synthesis and Optical Properties of ZnO Nanorods. Materials Science and Engineering: B, 121, 42-47. http://dx.doi.org/10.1016/j.mseb.2005.02.065
- Caglar, M., Ilican, S. and Caglar, Y. (2009) Influence of Dopant Concentration on the Optical Properties of ZnO: In Films by Sol-Gel Method. Thin Solid Films, 517, 5023-5028.
- Bao, D., Gu, H. and Kuang, A. (1998) Sol-Gel Derived C-Axis Oriented ZnO Thin Films. Thin Solid Films, 32, 47-39.
- Zhang, D.L., Zhang, J.B., Wu, Q.M. and Miao, X.S. (2010) Microstructure, Morphology, and Ultraviolet Emission of zinc Oxidenanocrystallin Films by the Modified Successive Ionic Layer Adsorption and Reaction Method. Journal of the American Ceramic Society, 93, 3284-3290.
- Khan, Z.R., Zulfequar, M. and Khan, M.S. (2010) Optical and Structural Properties of Thermally Evaporate Cadmium Sulfide Thin Films on Silicon (100) Wafer. Materials Science and Engineering: B, 174, 145-149.
- Foo, K.L., Hashim, U., Muhammad, K. and Voon, Ch.H. (2014) Sol-Gel Synthesized Zinc Oxide Nanorods and Their Structural and Optical Investigation for Optoelectronic Application. Nanoscale Research Letters, 429, 1-10.








