Graphene Sheets with Modified Surface by Sodium Lauryl Sulfate Surfactant for Biomedical Applications ()
1. Introduction
Graphene is one of allotropic modifications of carbon element with atomic number 6, located in the Periodic table of chemical elements, which was notable Nobel Prize in Physics by A. Geim and K. Novoselov [1] - [3] . Graphene’s structure is basically composed of monolayer carbon atoms hexagonal lattice, in which individual carbon atoms are connected by sp2 bonds, similar to those of nanotubes, fullerenes and carbon onions [4] [5] . Graphene is defining as two-dimensional (2D) material [6] [7] , due to its width and length characteristics. Recent progress has shown that the graphene-based materials can have a deep impact on electronic and optoelectronic devices, chemical sensors, nanocomposites and energy storage [8] . Fabrication of graphene was achieved by several methods: chemical reduction [9] , plasma let [10] , chemical vapor deposition [11] , and so on [12] .
Chemical modification of graphene dispersion by surfactants in the content of manufacturing and commercialization was reported [13] .
Sodium lauryl sulphate (SLS)-modified activated carbon from risk husk for waste lead (Pb) removal was synthesized by using of series of treatments, such as: carbonization, activation with H3PO4 and surface modification using sodium lauryl sulfate (SLS) [14] .
This work was aimed to synthesize graphene sheets with modified surface by sodium lauryl sulfate (SLS) surfactant, using the simple and one-pot solvothermal reaction method. Solvothermal method is based on a chemical reaction between initial substances, mainly, the solvent and dissolved substances in it. Solvothermal reaction is generated inside stainless steel autoclave, under temperature and pressure conditions, in definite duration of time. Surface functionalized graphene has a number of superior advantages for biomedical applications, in particular, manufacturing of attachments for polymer molecules, creating of novel biosurface with graphene, as reinforcement for biopolymer coatings such as ultra-high molecular weight polypropylene (UHMWPE), metallic biomaterials (Ti and Ti alloys) and bioceramics as hydroxyapatite (HA). For example, chemical functionalization of graphene enables this material to be processed by solvent assisted techniques, such as layer-by-layer assembly, spin-coating, and filtration [15] . Surface modification of graphene by SLS surfactant positively improves the attachment of polymer molecules for creating of novel biosurface with graphene [16] [17] . It is also known that surface modification of graphene reduces its toxicity in vitro and in vivo [18] . At the same time, synthesis and surface modification of graphene sheets by SLS surfactant using the one-step solvothermal method and characterizations of as-obtained graphene were described in this work.
2. Experimental Procedure
A schematics of solvothermal synthesis method is presented in the Figure 1. Stainless steel autoclave (capacity of 100 ml) fixed to the metal equipment body, and was connected with temperature controller. Initial substances (total volume 60 ml), composed of solvent (distilled water-ethanol mixture with 3:1 volume ratio), pure carbon powder 0.3 g (purity 99.9%, purchased from Kojundo Kagaku. Co.), sodium lauryl sulfate (SLS) surfactant (C12H25NaO4S, 5% solution, purchased from Kanto Chemical Co.) were placed inside the stainless steel autoclave and covered with lid. Solvothermal reaction temperature was set at 170˚C with further increase of inner-autoclave pressure to 1.4 MPa, and
![]()
Figure 1. Schematics of experimental setup: (a) Drawing of solvothermal synthesis equipment with labeled compartments: 1―vessel; 2―lid; 3―packing seal; 4―mount seal; 5―safety valve; 6―pressure gauge; 7―valve; 8―thermo well; 9―stirrer; 10―stirring blade; 11―stirrer cooling hose outlet; 12―wrench; 13―heater; 14―thermo sensor; 15―tempertaure controller; 16―rubber seal; 17―specimen pipe; 18―pressure safety branch compartment; (b) Figure of stainless steel autoclave containing initial reaction mixture; (c) Photograph of surface modified graphene sample obtained after solvothermal reaction.
reaction time lasted for 1 hour. Solvothermal synthesis experiment was conducted three times, with SLS surfactant amounts of: 1 ml, 3 ml and 5 ml, for determination of surfactant amount effect on the surface modification of graphene sheets. Graphene sheets with modified surface by SLS surfactant were characterized by high resolution transmission electron microscope (HRTEM) Technai F-20 S-Twin equipped with an Energy dispersive X-ray spectroscopy (EDX) detector for elemental analysis at 200 keV. The HRTEM samples were prepared by placing a drop of surface modified by SLS graphene sheets solution (in ethanol) on a holey carbon-coated copper grid. The excess solvent was evaporated and the specimen was dried in a vacuum overnight. X-ray photoelectron analysis was carried out on Thermo Scientific XPS spectrometer. Raman spectrum was collected on HORIBA Jobin Yvon HR800 spectrometer, with He-Ne laser at 516 nm. ATR-FTIR spectra were recorded using the JASCO FT/IR-6300 spectrometer. Atomic force microscopy analyses were conducted on 5500 AFM (N9410S) Keysight Technologies, Inc., with measurement mode: AAC (acoustic ac mode in air).
3. Results and Discussions
Multi layered graphene sheets formed after solvothermal reaction were detected by high resolution transmission electron microscope (HRTEM) and Raman spectroscopy analyses (Figure 2). HRTEM characterizations, which are proven to be an excellent tool for structure analysis [19] , gave us a lot of necessary information regarding the structure, shape location, morphology and sizes of observing materials. Solubilization of carbon powder in ethanol-water mixture at 170˚C during solvothermal reaction and further graphitization led to formation of graphene sheets. Sonication technique can be applied for synthesis of graphene and separation of graphene multi-layer sheets into single sheets [20] [21] , however, our focus was made on solvothermal synthesis and surface modification of graphene, rather than sonication and separation processes. Sonication technique can be applied for synthesis of graphene and separation of graphene multi-
![]()
Figure 2. (a) HRTEM photograph of multilayer graphene sheets synthesized by solvothermal reaction method; (b) Raman spectrum collected from graphene sheets synthesized by solvothermal method, by using He-Ne laser beam, inset corresponding to multilayer graphene, inset adapted from [28] by permission of Elsevier; (c) High resolution photograph of graphene at 1 nm scale, red square shows areas for revealed hexagonal honey-comb lattice structure of graphene, inset is enlarged area shown in red square in (c), corresponding to the inter-atomic distance of 0.142 nm between C-C covalent bonds; (d) FFT image taken parallel with HRTEM observation; (e) Peak profile showing inter-atomic distance between C-C bonds equal to 0.142 nm.
layer sheets into single sheets [22] [23] , however, our focus was made on solvothermal synthesis and surface modification of graphene, rather than sonication and separation processes. Similar to previously reported works [22] - [24] , solvothermally synthesized graphene exhibited analogical structures and dimensions. Inter-atomic distance between C-C bonds in solvothermally synthesized graphene are coincident with mechanically exfoliated graphene, were equal to 0.142 nm [25] [26] . Comparing to other works, solvothermally synthesized graphene was stable against oxidation, whereas surface modified graphene by various surfactants [27] [28] were undergo reduction and oxidation as well.
Raman spectroscopy was used to study the number of layers and structure of surface modified graphene sample synthesized by solvothermal method. Peaks for D band at 1327.29 cm−1, G band at 1572.5 cm−1 and second ordered 2D band at 2663.54 cm−1 are detected during Raman spectroscopy analysis. It can be seen that shape and intensity of Raman peaks can be different depending on laser and wavelength type used during analysis [29] . Also, D band in the Raman spectrum can be composed of more than one peak, such as D1 and D2, which is associated with scattering of electrons by mechanism of double resonance process [30] . Solvothermally synthesized graphene composed of multilayer, which can be explained by the intensity and shape of 2D band peak [31] [32] . The intensity of the G band increases with increased graphene layers, and the shape of 2D band evolves into four peaks of bilayer graphene [33] . The 2D peak in graphene is due to two phonons with opposite momentum in the highest optical branch near the K (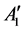 symmetry at K) [34] .
symmetry at K) [34] .
Figure 3 shows XPS spectrum collected from surface modified graphene sheets by solvothermal method, inset is C-C line C1s peak detected at 284.7 eV binding energy. Oxidation of graphene was not occurred during solvothermal reaction, which can be seen from the general survey, as there is only carbon atom C1s was appeared, and no oxygen atom peak was detected. Further qualification of obtained data is referred to [35] [36] .
ATR-FTIR spectroscopy was applied to determine SLS (C12H25NaO4S) surfactant molecules attached on the surface of graphene sheets. Figure 4 shows ATR-FTIR spec-
![]()
Figure 3. XRS spectra collected from surface modified by SLS surfactant graphene sheets: (a) Scan plot showing single peak for carbon C1s atom at 284.7 eV binding energy; (b) General survey showing peak for C1s carbon atom, and no peak for oxygen.
![]()
Figure 4. ATR-FTIR spectra of surface modified by SLS surfactant graphene sheets synthesized by solvothermal reaction method. Detected functional groups were released from SLS (C12H25NaO4S) surfactant during solvothermal reaction synthesis.
tra for pure carbon powder, surface modified graphene sheets by 1 ml SLS, and surface modified graphene sheets by 5 ml SLS, respectively. Adding of 5 ml SLS surfactant to the reaction mixture, provided higher intensity and much more number of peaks belonging to the ether (-S-OR) at 700 - 900 cm−1, thiocarbonyl (=C=S) at 1050 - 1200 cm−1 and sulfone (=S=O) at 1030 - 1450 cm−1 functional groups, respectively, in contrast to 1 ml of SLS surfactant amount.
Also, symmetric C-H stretches (CH3) at 2875 cm−1, asymmetric C-H stretches (CH2) at 2919 cm−1 and asymmetric C-H stretches (CH3) at 2956 cm−1 were detected. Attachment of above functional groups followed by release of them from SLS (C12H25NaO4S) surfactant during solvothermal reaction.
In comparison with other reported methods for surface modification of graphene, solvothermal reaction method have several advantages, such as: use of low temperature energy (1˚C - 300˚C), short reaction time (1 hour is quite enough), one-step process (synthesis of graphene and its surface modification can be achieved at the same time).
Atomic Force Microscopy is measurement technique, which can provide information not only about physical dimensions, but also let to detect surface features on nanomaterials. Figure 5 presenting AFM images of surface modified by SLS surfactant graphene sheets synthesized by solvothermal synthesis. Figure 5(a) shows solvothermally synthesized graphene sample which was dissolved in ethanol, dropped onto mica substrate and dried prior to observation. As it was discussed earlier [37] , SLS surfactant molecules attached onto graphene surface by using of low energy solvothermal reaction method, were observed as protuberant areas on the surface of graphene flakes shown in the Figure 5(a) inside red square, with height profile indication of 4.8 nm. Figure 5(c) shows surface modified graphene sheets by SLS surfactant, when sample was subjected onto HOPG substrate for AFM observation. Line profile in panel (d) represents the average obtained within the yellow line in panel (c), and the virtual nanosheets ≈ 27 nm
![]()
Figure 5. AFM images of surface modified by SLS surfactant graphene sheets synthesized by solvothermal method: (a) AFM image taken by using the mica substrate, inset is enlarged area shown in small red square, arrows indicating surfactant molecules attached to the graphene surface; (b) Height profile projected within the yellow line and showing graphene sheets height equal to 4.8 nm; (c) AFM image of SLS surfactant modified graphene sheets taken using the HOPG as substrate; (d) Line profile obtained within the yellow line in panel (c), and shows graphene nanosheets height attached with surfactant, equal to 27 nm.
high (relative to HOPG substrate).
Table 1 presenting Antibacterial activity of surface modified by SLS surfactant graphene sheets, studied using 3 M Petrifilms for E. coli bacterial colonies. It was observed, that antibacterial activity was higher for graphene sheets utilized 5 ml of SLS for surface modification, comparing to samples utilized 3 ml and 1 ml of SLS surfactant. Following results were obtained after average E. coli bacteria colonies calculation: for case 136.6 μg/ml of surface modified by SLS graphene nanosheets, 660 counts for 1 ml of SLS utilization, 570 counts for 3 ml of SLS utilization, and 376 counts for 5 ml of SLS surfactant utilized for surface modification. Antibacterial activity can be initiated by inhibition of metabolic processes [38] in E. coli bacterium colonies.
Summary of graphene family materials discussed [39] , indicating slight and nearly no toxic effects of graphene, reduced graphene and oxidized graphene against E. coli bacterium colonies.
These obtained results are certainly supporting the biomedical applications of surface
![]()
Table 1. Antibacterial activity of surface modified by SLS surfactant graphene sheets synthesized by solvothermal reaction method.
modified by SLS surfactant graphene nanosheets. Graphene elicit toxic effects both in vitro and in vivo, whereas surface modifications can significantly reduce its toxic interactions with living systems [17] , which allows biomedical applications of surface modified by SLS graphene sheets, in particular as inner linings and as reinforcement for biopolymer coatings such as ultra-high molecular weight polypropylene (UHMWP), Ti and Ti alloys and hydroxyapatite (HA).
4. Conclusion
Synthesis of surface modified graphene sheets by SLS surfactant during solvothermal reaction was highlighted in this work. Atomic state and elemental composition of surface modified graphene were consisted of C-C bonds with inter atomic distance 0.124 nm, and associated with C1s XPS peak, excluding oxidation of synthesized graphene. Multilayer graphene sheets consisting of hexagonal honey-comb lattice were revealed by HRTEM, and structure was determined by Raman spectroscopy. Influence of SLS surfactant amount on surface modification and attached functional groups were determined by attenuated total reflection FTIR spectroscopy. Higher amount of surfactant resulted in much more surface modification areas with higher intensity peaks and quantities of functional groups. Antibacterial activity of surface modified by SLS surfactant graphene sheets is found to be higher in case of increased amount SLS surfactant used during surface modification. Obtained results are highlighting biomedical applications of surface modified by SLS graphene sheets, as biocompatible coating platform between polymer, metallic, ceramic type implants and the living tissue.
Acknowledgements
This work was funded by start-up research acceleration fund (Funding number: 535- 11535016) of the Kumamoto University.