 Journal of Cancer Therapy, 2011, 2, 302-307 doi:10.4236/jct.2011.23040 Published Online August 2011 (http://www.SciRP.org/journal/jct) Copyright © 2011 SciRes. JCT The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer Inès Ayadi Masmoudi1, Amine Masmoud i1, Nabil Toumi1, Afef Khanfir1, Mounir Frikha1, Jamel Daoud2 1Department of Medical Oncology; 2Department of Radiation Oncology, Habib Bourguiba University Hospital, Sfax, Tunisia. Email: inesmaj@yahoo.fr Received May 3rd, 2011; revised June 22nd, 2011; accepted July 1st, 2011. ABSTRACT Obesity is associated with worse breast cancer outcomes in Western and Asian women. The present study was to deter- mine the relation of body mass index (BMI) to disease-free survival (DFS) in Tunisian patients with operable breast cancer. We retrospectively reviewed data from 227 patients with operable breast cancer treated with primary surgery and adjuvant epirubicin-based chemotherapy. Patients were categorized into lower-BMI (<27.5 kg/m2; N = 114) v higher-BMI (>27.5 kg/m2; N = 113). Patients’ and disease characteristics, including tumor size and lymph node status, were similar between the 2 groups. With a median follow-up of 56 months, there were 42 and 30 DFS events in the lower- and higher-BMI group, respectively (adjusted hazard ratio (HR): 0.74; 95% CI, 0.43 to 1.26; p = 0.26). A sig- nificant interaction (p < 0.001) was found between BMI category and time, with a significant DFS advantage for the higher-BMI group after 3 years (adjusted HR: 0.28; 95% CI, i to 0.86; p = 0.03). We conclude that overweight is asso- ciated with a decreased risk of late recurrence in Tunisian patients with operable breast cancer. Keywords: Adjuvant Chemotherapy, Early Breast Cancer, Obesity, Recurrence, Tunisia 1. Introduction In Tunisia, breast cancer is the leading female cancer with an adjusted incidence of 30 per 100,000 per year [1]. Although patients with operable disease account for ap- proximately two thirds of the cases, they usually present with large tumors and/or lymph node involvement [2], thus requiring a multimodality therapeutic approach. In another hand, overweight, defined by a body mass index (BMI) higher than 25 kg/m2, is a common problem among Tunisian women, with a prevalence of 51%, and the half of them being obese (BMI > 30 kg/m2) [3]. Dur- ing the last two decades, a growing amount of literature had demonstrated a negative impact for overweight at diagnosis on the outcome of breast cancer [4]. The prog- nostic value of high BMI was consistently reported in various settings, including post-menopausal and pre- menopausal patients, early stage and locally-advanced disease, and different ethnic groups, mainly Caucasians and Asians [5-9]. Whether such correlation exists in North-African women with breast cancer is not docu- mented. In this context, the present study aimed to investigate the relation of BMI to the outcome in a cohort of Tuni- sian women with operable breast cancer uniformly treated with primary surgery followed by epirubicin- based adjuvant chemotherapy. 2. Patients and Methods Between January 1995 and December 2005, 322 patients with operable breast carcinoma received adjuvant epiru- bicin-based chemotherapy in our centre. Information on weight and height at diagnosis was available for 227 pa- tients, who form the basis for this retrospective study. Operable breast cancer was defined as clinical T0-3N0-1 or T4b N0-1 disease with limited skin in- volvement. A baseline negative staging work-up includ- ing chest x-ray, liver ultra sonography and bone scan was mandatory for all patients, regardless of clinical stage. Primary surgery consisted of radical modified mastec- tomy or breast-sparing operation, with axillary lymph node dissection. All patients received adjuvant chemotherapy, which  The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer303 was mandated by the presence of one or more of the fol- lowing risk factors: age <35, pathological tumor size >2 cm, lymph node involvement, negative hormone receptor status, histological grade 2 or 3. Radiotherapy was indi- cated after chemotherapy in the presence of any of the following: breast-conserving surgery, lymph node in- volvement, clinical stage T3 or T4b, multifocal/multi- center tumor, or microscopic positive margins (R1 resec- tion). For patients with hormone-receptor positive dis- ease, tamoxifen 20 mg daily was given regardless of menopausal status. BMI at diagnosis, calculated as the weight (kg) di- vided by the height (m) squared, was dichotomised above and below the median value. The comparison between higher-BMI (BMI-H) and lower-BMI (BMI-L) groups was performed using Wilcoxon rank sum test for con- tinuous variables, and khi-square or Fisher exact test, as appropriate, for categorical variables. Variables with p-values < 0.20 were considered of borderline signifi- cance and were included as covariates for subsequent analyses. We used the Kaplan-Meier method to compute survival plots. DFS was defined as the time from surgery until relapse (local or distant), contralateral breast cancer or death, whichever occurred first. OS was calculated from surgery until death from any cause or last follow-up. We performed a Cox regression that adjusted for pa- tients’ characteristics that were different (p < 0.20) be- tween the two groups to calculate hazard ratios (HR) for DFS between BMI-H and BMI-L groups. To test the interaction between BMI category and time, we com- pared the effect of BMI on DFS in two consecutive time intervals using a piecewise Cox regression model. 3. Results 3.1. Patients’ and Disease Characteristics Median age at diagnosis was 48 (range: 24 - 75). Among 162 patients for whom findings from initial physical examination were known, the majority had stage II or stage III disease (65% and 32%, respectively). Among 216 (97%) patients with available information for hor- mone receptor status, estrogen and/or progesterone re- ceptors were positive in 134 (62%) of the cases. As it can be seen in Table 1, most patients had histologic grade 2 or 3 (87%) ductal infiltrating carcinoma (86%), with a tumor size >2 cm (70%) and lymph node involvement (60%). According to the World Health Organisation (WHO) classification, 85 patients had grade 1 overweight (BMI between 25 and 29.9 kg/m2) and 76 were obese (BMI ≥ 30 kg/m2), for a total overweight rate of 71%. Sixty two (27%) patients were in the normal BMI range, while the remaining 4 (2%) patients were in the lowest BMI cate- Table 1. Patients and disease characteristics. BMI <27.5 kg/m2 (N = 114) N (%) ≥27.5 kg/m2 (N = 113) N (%) p-value Age Mean 48 50 0.193¶ ≤34 11 (10) 7 (6) 35 - 49 56 (49) 50 (44) ≥50 47 (41.2) 56 (50) Menopausal status* 0.017 Pre/peri-menopausal62 (61) 44 (44) Post-menopausal 40 (39) 56 (56) Unknown 12 13 Histological type 0.803 Infiltrating ductal 99 (87) 97 (86) Infiltrating lobular 6 (5) 7 (6) Mixed carcinoma 6 (5) 4 (4) Other 3 (3) 5 (4) Tumor size (pT)* 0.359 ≤2 cm 37 (34) 28 (26) >2 cm but ≤5 cm 59 (55) 68 (64) >5 cm 12 (11) 10 (9) Unknown 6 7 Positive nodes 0.204 0 41 (36) 50 (44) 1 to 3 41 (36) 42 (37) ≥4 32 (28) 21 (19) Histologic grade* 0.219 1 11 (11) 16 (15) 2 57 (54) 46 (43) 3 37 (35) 46 (43) Unknown 9 5 Hormone receptors* 0.115 Positive 62 (57) 72 (67) Negative 47 (43) 35 (33) Unknown 5 6 Type of surgery 0.394 Mastectomy 93 (82) 87 (77) Breast-sparing 21 (18) 26 (23) Chemotherapy 0.404 FEC50/60 78 (68) 83 (74) FEC100 36 (32) 30 (27) Radiotherapy 0.683 Yes 99 (87) 96 (85) No 15 (13) 17 (15) Endocrine therapy 0.518 Tamoxifen 40 (58) 41 (53) Tamoxifen then AI 26 (38) 31 (40) Up-front AI 0 (0) 2 (3) OS/OA alone 2 (3) 1 (1) None 1 (1) 3 (4) ¶ Wilcoxon rank sum test; * Percentages were calculated among patients for whom information on studied variable was available, Abbreviation: AI: aromatase inhibitor, OS/OA: ovarian suppression/ ovarian ablation gory (<18.5 kg/m2). Median BMI value was 27.5 kg/m2. Compared with those whom BMI was above the median, leaner patients tended to be younger, more likely to be pre- or peri-meno- pausal, and more likely to have hormone-unresponsive tumors (0.05 < p < 0.20). Other disease characteristics, including tumor size and lymph node involvement, did not significantly differ between the two groups (Table 1). Copyright © 2011 SciRes. JCT  The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer 304 3.2. Treatment Surgery consisted of total mastectomy in the majority (79%) of the patients. Radiotherapy was given in 88% and 100% of the cases after mastectomy and breast- sparing surgery, respectively. All patients underwent fluoro-uracil, epirubicin and cyclophosphamide (FEC) adjuvant chemotherapy, with varying epirubicin doses (50, 60 or 100 mg/m2). No statistically-significant dif- ferences were found between the two BMI groups with respect to treatment modalities (Table 1). 3.3. Survival Outcomes At a median follow-up of 56 months, 5-year DFS for the entire cohort was 70%. DFS and OS events in each BMI group are summarised in Table 2. There was a non-statistically significant 6% absolute difference between the two BMI groups in terms of DFS, with a 5-year DFS of 73% and 67% for the BMI-H and BMI-L groups, respectively (HR for BMI-H versus BMI-L: 0.72; 95% CI, 0.45 to 1.15; log-rank p = 0.17) (Figure 1(a)). Comparable results were found after ad- justing for age, menopausal status and hormone-receptor status (HR = 0.70; 95% CI, 0.41 to 1.18). The rate of overall survival (OS) at 5 years was 82% in the higher-BMI group and 77% in the lower-BMI group (HR = 1.00, 95% CI, 0.57 to 1.79; log-rank p = 0.98) (Figure 1(b)). On the basis on Figure 1(a), we analysed DFS using a piecewise Cox regression with change point at 3 years. There was a statistically significant interaction between BMI category and time (p < 0.001), implying that the effect of BMI changes across time. Within the first 3 years, the adjusted HR for DFS between BMI-H and BMI -L groups was 1.13 (95% CI, 0.59 to 2.15). After 3 years, the likelihood of a DFS event was nearly 4 fold lower for the BMI-H group compared with the BMI-L group (log-rank p = 0.03; adjusted HR = 0.28; 95% CI, 0.09 to 0.84). The unadjusted HR for DFS between BMI-H and BMI-L groups in various patients subgroups are shown in Table 3. No significant interaction was found between BMI and any of the patients’ and disease characteristics analysed (p values not shown). 4. Discussion Unexpectedly, the present study shows that overweight is not associated with advanced tumor stage or lymph node involvement at diagnosis in Tunisian women with oper- able breast cancer. Moreover, a major finding from our study is that overweight is associated with a reduced risk of late recurrence. In view of the existing evidence dem- onstrating a negative effect of overweight on breast can- Table 2. Disease-free survival events summary. BMI < 27.5 kg/m2 (N = 114) BMI > 27.5 kg/m2 (N = 113) Recurrence Distant only 25 19 Both distant and local 11 7 Local only 5 2 Contralateral 1 1 Death After recurrence 25 22 Without previous recurrence 0 1 Total events 42 30 Table 3. Hazard ratios for breast cancer recurrence by body mass index category . Overall >3 years HR*95% CI HR* 95% CI Age <50 0.810.45 - 1.45 0.54 0.18 - 1.59 ≥50 0.600.27 - 1.35 0.22 0.05 - 1.01 Menopausal status Pre/peri-menopausal0.830.43 - 1.60 0.49 0.13 - 1.80 Post- menopausal 0.590.24 - 1.43 0.12 0.01 - 1.00 Hormone receptors Positive/unknown 0.640.32 - 1.27 0.39 0.12 - 1.25 Negative 0.920.47 - 1.78 0.45 0.12 - 1.71 Tumor size ≤2 cm 0.370.08 - 1.73 0.43 0.16 - 1.17 >2 cm 0.700.41 - 1.21 0.31 0.04 - 2.63 Lymph Node status Negative 0.600.23 - 1.59 0.14 0.02 - 1.20 Positive 0.840.48 - 1.44 0.61 0.23 - 1.62 Histologic grade Grade ½ 0.640.32 - 1.26 0.45 0.14 - 1.42 Grade 3 0.750.36 - 1.55 0.36 0.09 - 1.43 *Unadjusted hazard ratio of higher- to lower-body mass index. Abbre- viation: HR, hazard ratio. cer patients’ DFS and OS, our results are seemingly sur- prising. In view of reported association between increased body weight at diagnosis and advanced disease, it has been hypothesised that the higher exposure to endoge- nous estrogens mediated by adipose tissue may explain the prognostic effect of obesity. In our study, tumor stage and lymph node involvement were similar between BMI-H and BMI-L groups. One may argue that the dif- ference in DFS may be due to the fact that more BMI-H patients had hormone-receptor positive disease. However, the effect of overweight was not modified after adjust- ment for hormone receptor status. It has been suggested that the effect of obesity on breast cancer outcomes may be mediated by leptin, a neuroen- docrine hormone linked to obesity [10]. However, ele- vated levels of leptin were not associated with outcome in 471 women with operable breast cancer [11]. Recently, a Tunisian group investigated the prognostic value of polymorphisms in leptin gene 5’ promoter region, Copyright © 2011 SciRes. JCT 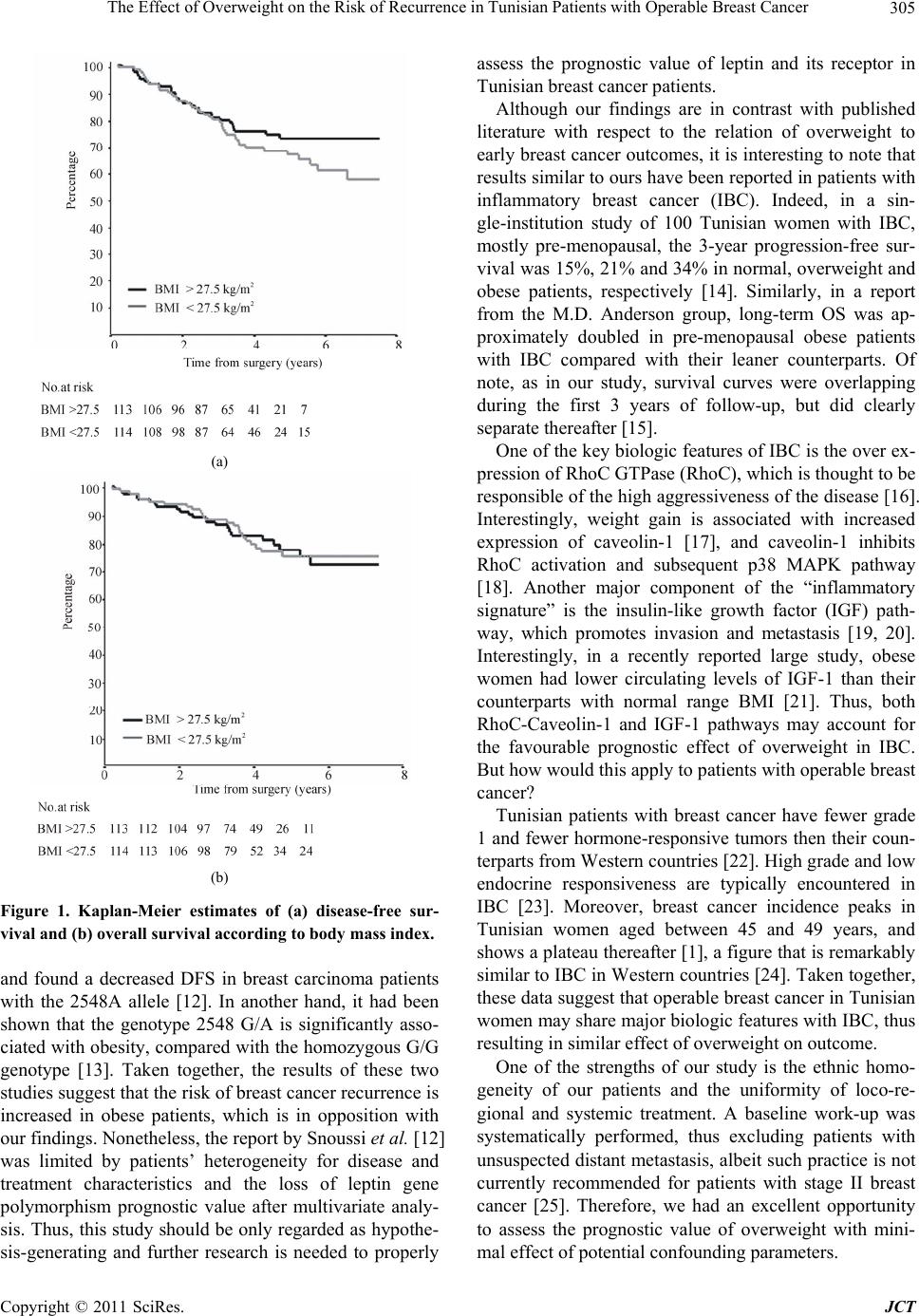 The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer305 (a) (b) Figure 1. Kaplan-Meier estimates of (a) disease-free sur- vival and (b) overall survival according to body mass index. and found a decreased DFS in breast carcinoma patients with the 2548A allele [12]. In another hand, it had been shown that the genotype 2548 G/A is significantly asso- ciated with obesity, compared with the homozygous G/G genotype [13]. Taken together, the results of these two studies suggest that the risk of breast cancer recurrence is increased in obese patients, which is in opposition with our findings. Nonetheless, the report by Snoussi et al. [12] was limited by patients’ heterogeneity for disease and treatment characteristics and the loss of leptin gene polymorphism prognostic value after multivariate analy- sis. Thus, this study should be only regarded as hypothe- sis-generating and further research is needed to properly assess the prognostic value of leptin and its receptor in Tunisian breast cancer patients. Although our findings are in contrast with published literature with respect to the relation of overweight to early breast cancer outcomes, it is interesting to note that results similar to ours have been reported in patients with inflammatory breast cancer (IBC). Indeed, in a sin- gle-institution study of 100 Tunisian women with IBC, mostly pre-menopausal, the 3-year progression-free sur- vival was 15%, 21% and 34% in normal, overweight and obese patients, respectively [14]. Similarly, in a report from the M.D. Anderson group, long-term OS was ap- proximately doubled in pre-menopausal obese patients with IBC compared with their leaner counterparts. Of note, as in our study, survival curves were overlapping during the first 3 years of follow-up, but did clearly separate thereafter [15]. One of the key biologic features of IBC is the over ex- pression of RhoC GTPase (RhoC), which is thought to be responsible of the high aggressiveness of the disease [16]. Interestingly, weight gain is associated with increased expression of caveolin-1 [17], and caveolin-1 inhibits RhoC activation and subsequent p38 MAPK pathway [18]. Another major component of the “inflammatory signature” is the insulin-like growth factor (IGF) path- way, which promotes invasion and metastasis [19, 20]. Interestingly, in a recently reported large study, obese women had lower circulating levels of IGF-1 than their counterparts with normal range BMI [21]. Thus, both RhoC-Caveolin-1 and IGF-1 pathways may account for the favourable prognostic effect of overweight in IBC. But how would this apply to patients with operable breast cancer? Tunisian patients with breast cancer have fewer grade 1 and fewer hormone-responsive tumors then their coun- terparts from Western countries [22]. High grade and low endocrine responsiveness are typically encountered in IBC [23]. Moreover, breast cancer incidence peaks in Tunisian women aged between 45 and 49 years, and shows a plateau thereafter [1], a figure that is remarkably similar to IBC in Western countries [24]. Taken together, these data suggest that operable breast cancer in Tunisian women may share major biologic features with IBC, thus resulting in similar effect of overweight on outcome. One of the strengths of our study is the ethnic homo- geneity of our patients and the uniformity of loco-re- gional and systemic treatment. A baseline work-up was systematically performed, thus excluding patients with unsuspected distant metastasis, albeit such practice is not currently recommended for patients with stage II breast cancer [25]. Therefore, we had an excellent opportunity to assess the prognostic value of overweight with mini- mal effect of potential confounding parameters. Copyright © 2011 SciRes. JCT  The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer 306 Nonetheless, as with any study, our study has some limitations. The relatively small number of patients and short follow-up made it difficult to detect a difference in OS between BMI groups. Moreover, our analysis was based on BMI dichotomization on its median rather than WHO-defined BMI categories. However, the relevance of WHO-defined BMI categories to non-European popu- lations has been questioned [26], and remains to be proved in North African population. 5. Conclusions A negative impact for overweight at diagnosis on the outcome of breast cancer has been demonstrated in the literature. In our study, we found an inverse correlation between overweight and the risk of recurrence in Tuni- sian women with operable breast cancer. Another finding in contrast with previous literature was the lack of asso- ciation between obesity and disease stage at presentation, reinforcing the hypothesis of a biologically different dis- ease. International partnership is urgently needed to promote research aiming to study the molecular profile and identify cell pathways involved in breast carcino- genesis in Tunisian women. REFERENCES [1] M. Ben Abdallah, S. Zehani, M. Maalej, M. Hsairi, M. Hechiche, K. Ben Romdane, H. Boussen, A. Saadi, N. Achour and F. Ben Ayed, “Cancer du sein en Tunisie : Carctéristiques Epidémiologiques et Tendance Evolutive de l’Incidence, ” Tunisie Médicale, Vol. 87, No. 7, 2009, pp. 417-425. [2] A. Khanfir, M. Frikha, F. Kallel, M. Meziou, K. Trabelsi, T. Boudawara, J. Mnif and J. Daoud, “Le Cancer du sein de la Femme Jeune Dans le sud Tunisien,” Cancer Radiother, Vol. 10, No. 8, 2006, pp. 565-571. doi:10.1016/j.canrad.2006.09.115 [3] N. Mokhtar, J. Elati, R. Chabir, A. Bour, K. Elkari, N. P. Schossman, B. Caballero and H. Aguenaou, “Diet Culture and Obesity in Northern Africa,” Journal of Nutrition, Vol. 131, No. 3, 2001, pp. 887S-892S. [4] J. J. Dignam and E. P. Mamounas, “Obesity and Breast Cancer Prognosis: An Expanding Body of Evidence,” Annals of Oncology, Vol. 15, No. 6, 2004, pp. 850-851. doi:10.1093/annonc/mdh241 [5] International Breast Cancer Study Group, G. Berclaz, S. Li, K. N. Price, A. S. Coates, M. castiglione-Gertsch, C. M. Denstam, S. B. Holmberg, J. Lindtner, D. Erzen, J. Collins, R. Synder, B. Thurlimann, M. F. Fey, C. Men- diola, I. D. Werner, E. Simoncini, D. Crivellari, R. D. Gelber and A. Goldhirsch, “Body Mass Index as a Prog- nostic Feature in Operable Breast Cancer: The Interna- tional Breast Cancer Study Group Experience,” Annals of Oncology, Vol. 15, No. 6, 2004, pp. 875-884. doi:10.1093/annonc/mdh222 [6] S. Loi, R. L. Milne, M. L. Friedlander, M. R. E. McCre- die, G. G. Giles, J. L. Hopper and K. A. Phillipps, “Obe- sity and Outcomes in Premenopausal and Postm-enopausal Breast Cancer,” Cancer Epidemiology, Biomarkers & Prevention, Vol. 14, No. 7, 2005, 1686-1691. pp. doi:10.1158/1055-9965.EPI-05-0042 [7] P. E. Abrahamson, M. D. Gammon, M. J. Lund, E. W. Flagg, P. L. Porter, J. Stevens, S. A. Swanson, L. A. Brinton, J. W. Eley and R. J. Coates, “General and Ab- dominal Obesity and Survival among Young Women with Breast Cancer,” Cancer Epidemiology, Biomarke & Prevention, Vol. 15, No. 10, 2006, p rs p. 1871-1877. doi:10.1158/1055-9965.EPI-06-0356 [8] S. Dawood, K. Broglio, A. M. Gonzalez-Angulo, S. W. Kau, R. Islam, G. N. Hortobagyi and M. Cristofanilli, “Prognostic Value of Body Mass Index in Locally Ad- vanced Breast Cancer,” Clinical 14, No. 6, 2008, pp. 1718-1725. Cancer Research, Vol. doi:10.1158/1078-0432.CCR-07-1479 [9] M. H. Tao, X. O. Shu, Z. X. Ruan, Y. T. Gao and W. Zheng, “Association of Overweight with Breast Cancer Survival,” American Journal of Epidemiology, Vol. 163, No. 2, 2006, pp. 101-107. doi:10.1093/aje/kwj017 [10] D. P. Rose, E. M. Gilhooly and D. W. Nixon, “Adverse Effects of Obesity on Breast Cancer Prognosis, and the Biological Actions of Leptin (Review),” International Journal of Oncology, Vol. 21, No. 6, 2002, pp. 1285- . 25, 2005, pp. 1292. [11] P. J. Goodwin, M. Ennis, I. G. Fantus, K. I. Pritchard M. E. Trudeau, J. Koo and N. Hood, “Is Leptin a Mediator of Adverse Prognostic Effects of Obesity in Breast Cancer?” Journal of Clinical Oncology, Vol. 23, No 6037-6042. doi:10.1200/JCO.2005.02.048 [12] K. Snoussi, A. D. Strosberg, N. Bouaouina, S. Ben Ah- med, A. N. helal and L. chouchane, “Leptin and Leptin Receptor Polymorphisms Are Associated with Increased Risk and Poor Prognosis of Breast Carcinoma,” BMC S1, 2009, pp. Cancer, Vol. 20, 2006, pp. 6-38. [13] M. F. Mnif, A. Zouari, S. Sessi, M. Elleuch, M. Fourati, K. Makni, N. Charfi and M. Abid, “Association du Polymorphisme G-2548 A de la Région 5 du Gène de la Leptine avec l’Obésité dans la Région de Sfax en Tunisie,” Diabetes Metabolism, Vol. 35, No. 56-57. doi:10.1016/S1262-3636(09)71920-2 [14] S. I. Labidi, K. Mrad, A. Mezlini, M. Ayadi Warda, J. D. Coombs, M. Ben Abdallah, K. Ben Romdhane, P. Viens and F. Ben Ayed, “Inflammatory Breast Cancer in Tuni- sia in the Era of Multimodality Therapy,” A cology, Vol. 19, No. 3, 2008, p nnals of On- p. 473-480. doi:10.1093/annonc/mdm480 [15] S. Chang, J. R. Alderfer, L. Asmar and A. U. Buzdar, “Inflammatory Breast Cancer Survival: The Role of Obe- sity and Menopausal Status at Diagnosis,” Breast Cancer Research and Treatment, Vol. 64, No. 2, 2000, pp. 157- 163. doi:10.1023/A:1006489100283 [16] K. L. Van Golen, Z. F. Wu, X. T. Qiao, L. W. Bao and S. D. Merajver, “RhoC GTPase, a Novel Transforming On- cogene for Human Mammary Epithelial Cells That Par- Copyright © 2011 SciRes. JCT  The Effect of Overweight on the Risk of Recurrence in Tunisian Patients with Operable Breast Cancer Copyright © 2011 SciRes. JCT 307 r Research, Vol. 60, No. 20, 2000, pp. eigh Cave- l. 4, 2005, pp. 4-21. tially Recapitulates the Inflammatory Breast Cancer Phe- notype,” Cance 5832-5838. [17] P. Singh, T. E. Peterson, A. Romero-Corral, D. E. Davidson, M. D. Jensen and V. K. Somers, “Identifica- tion of Caveolin-1 as an Inhibitor of Leptin Signaling: Implications for Leptin Resistance in a Human Wt Gain Model,” Circulation, Vol. 118, 2008, p. S278. [18] M. Lin, M. M. DiVito, S. D. Merajver, M. Boyanapalli and K. L. Van Golen, “Regulation of Pancreatic Cancer Cell Migration and Invasion by RhoC GTPase and olin-1,” Molecular Cancer, Vo doi:10.1186/1476-4598-4-21 [19] S. Van Laere, I. Van der Auwera, G. Van den Eynden, P. Van Hummelen, P. Van Dam, E. Van Marck, P. B. Vermeulen and L. Dirix, “Distinct Molecular Phenotype of Inflammatory Breast Cancer Compared to Non-In- flammatory Breast Cancer Using Affymetrix-Based Ge- nome-Wide Gene-Expression Analysis,” British of Cancer, Vol. 97, No. 8, 20 Jo 07, pp. 1165-1174. urnal doi:10.1038/sj.bjc.6603967 [20] X. Zhang, M. Lin, K. L. Van Golen, K. Yoshioka, K. Itoh and D. Yee, “Multiple Signalling Pathways Are Activated during Insulin-Like Growth Factor-I (IGF-I) Stimulated Breast Cancer Cell Migration,” Breast Cancer Re and Treatment, Vol. 93, No. 2, 2005, pp search . 159-168. doi:10.1007/s10549-005-4626-8 [21] I. T. Gram, T. Norat, S. Rinaldi, L. Dossus, A. Lukanova, B. Tehard, F. Clavel-Chapelon, C. H. Van Gils, P. A. H. Van Noord, P. H. M. Peeters, H. B. Bueno-De-Mesquita, G. Nagel, J. Linseisen, P. H. Lahamann, H. Boeing, D. Palli, C. Sacerdote, S. Panico, R. Tumino, S. Sieri, M. Dorronsoro, J. R. Quiros, C. A. Navarro, A. Barricarte, M. J. Tormo, C. A. Gonzalez, K. Overvad, S. P. Johnsen, A. Olsen, A. Tjonneland, R. Travis, N. Allen, S. Bingham, K. Riboli and R. Kaaks, “Body Mass Index, Waist Cir- cumference and Waist-Hip Ratio and Serum Levels of IGF-I and IGFBP-3 in European Women,” International Journal of Obesity (Lond), Vol. 30, No. 11, 2006, pp. 1623-1631. T. Khaw, P. Stattin, A. Trichopoulou, C. Casagrande, E. doi:10.1038/sj.ijo.0803324 [22] L. Ayadi, A. Khabir, H. Amouri, S. Karray, A. Dammak, M. Guermazi and T. Boudawara, “Correlation of HER-2 over-Expression with Clinico-Pathological Parameters in Tunisian Breast Carcinoma,” World Journal of Surgical Oncology, Vol. 6, No. 1, 2008, p. 112. doi:10.1186/1477-7819-6-112 [23] E. Charafe-Jauffret, K. Mrad, S. Labidi, A. Ben Hamida, K. Ben Romdhane, M. Ben Abdallah, C. Ginestier, B. Esterni, D. Birnbaum, F. Ben Ayed, L. Xerri, A. Mezlini and J. Jacquemier, “Inflammatory Breast Cancers in Tu- nisia and France Show Similar Immunophenotypes,” Breast, Vol. 16, No. 4, 2007, pp. 352-358. doi:10.1016/j.breast.2007.01.002 [24] K. W. Hance, W. F. Anderson, S. S. Devesa, H. A. Young and P. H. Levine, “Trends in Inflammatory Breast Carcinoma Incidence and Survival: The Surveillance, Epidemiology, and End Results Program at the National Cancer Institute,” Journal of the National Cancer Insti- tute, Vol. 97, No. 13, 2005, pp. 966-975. doi:10.1093/jnci/dji172 [25] F. Puglisi, A. Follador, A. M. Minisini, G. G. Cardellino, S. Russo, C. Andreetta, S. Di Terlizzi and A. Piga, “Base- line Staging Tests after a New Diagnosis of Breast Cancer: Further Evidence of Their Limited Indications,” Annals of Oncology, Vol. 16, No. 2, 2005, pp. 263-266. doi:10.1093/annonc/mdi063 [26] S. Low, M. C. Chin, S. Ma, D. Heng, M. Deurenberg-Yap, et al., “Rationale for Redefining Obesity in Asians,” An- nals Academy of Medicine Singapore, Vol. 38, No. 1, 2009, pp. 66-69.
|