1. INTRODUCTION
The number of patients requiring and receiving biomedical implants to correct defects and heal diseases of the skeletal system is constantly increasing [1]. Tissue engineering and regenerative medicine assist with restoration of damaged tissues using both functional cells and biodegradable scaffolds made from engineered biomaterials [2,3]. Significant efforts in the last decades have been devoted to the fabrication of porous and highly interconnected bioceramic scaffolds based on hydroxyapatite (HA), tricalcium phosphate and related composite materials for application in bone tissue engineering [4-6].
In this regard, Bioglass® 45S5 characterised with high amounts of Na2O and CaO, and relatively high CaO/P2O5 ratio [7] has shown capacity to induce favourable intracellular and extracellular responses promoting rapid bone formation [8-11] through formation of a hydroxycarbonate apatite (HCA) layer.
The major approaches to processing bioactive glass materials include; the melting and the sol-gel methods. The melting method utilizes high temperature in the range of 1300˚C - 1400˚C, while a major advantage of the sol-gel method is the much lower processing temperature, coupled with the ability to control the compositional range of the glass [6].
A major limitation of bioactive glasses, especially in the porous form is that they display low mechanical properties compared to cortical and cancellous bone [12,13], thus restricting their wide range of applications.
Some previous efforts [14,15] have shown that when sintered under optimal conditions, sodium containing bioglass attains nearly full densification and produces fine crystals of Na2Ca2Si3O9 having high bioactivity index [16] to give scaffolds with competent mechanical strength.
The serious challenge to the large scale preparation of bioactive glasses and glass-ceramic materials is the high cost of the widely used alkoxide precursors such as; tetraethyl orthosilicate (TEOS) and tetramethyl orthosilicate (TMOS) [17-20]. Consequently, some authors like Nayak et al. [21], Wu et al. [22] and Zanotto et al. [23] have explored alternative silica sources in their pathway to large scale bioactive glasses.
Clay minerals are fine grained hydrous silicates of octahedral or tetrahedral layered arrangement. Their chemical composition implies that they may yet serve as an economically viable non-toxic silica source for large scale synthesis of bioactive glasses.
Bentonite clay; (Na,Ca)(Al,Mg)6(Si4O10)3(OH)6∙nH2O formed from the weathering of volcanic ash is naturally occurring sedimentary clay with the 3-layered clay structure. It displays one aluminium oxide sheet sandwiched by two silicon oxide sheets Figure 1 [24]. Structurally, the internal aluminium and external silicon oxide sheet share oxygen atoms, while Mg2+ ions often substitute for Al3+ ions resulting in net negative charge [25]. The charge in the platelet is balanced by counter ions, usually
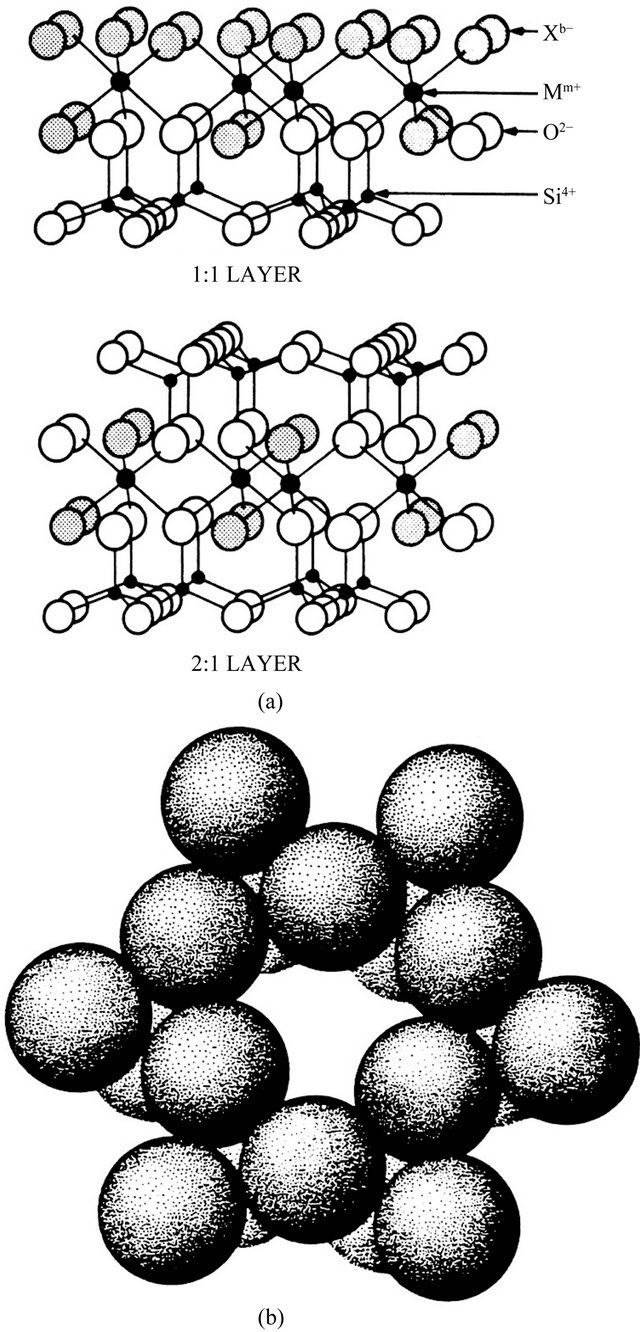
Figure 1. Crystal structure of clay mineral showing (a) 1:1 and 2:1 layer type clay, where X (shaded circles) is usually OH and M can be Al, Mg, Fe, etc; (b) Siloxane cavity in the basal plane of a tetrahedral sheet [24].
Na+ and Ca2+ located between the platelets. Clay has a wide origin and is particularly cheap and widely available in Nigeria [26]. The silicon oxide layer can be extracted using alkali at elevated temperature to give the corresponding alkali silicate as possible precursor for glass synthesis.
In this research work, we focus on the use of sodium bentonite as a source of silica to prepare sodium containing bioceramic material similar in composition to Bioglass® 45S5 quaternary system via the sol-gel process. To the best of our knowledge the use of bentonite clay as a precursor for this type of bioceramic has not been previously reported in literature.
2. MATERIALS AND EXPERIMENTAL PROCEDURES
2.1. Materials
The following chemicals were used as obtained for the preparation of the bioceramic material; bentonite clay- (OH)4Si8Al4O20∙nH2O (obtained locally), nitric acid (Fluka, Germany), phosphorus pentoxide (BDH Laboratory, 98%), sodium nitrate (Sigma-Aldrich, 99%), and calcium nitrate tetrahydrate (LOBA, 98%).
2.2. Experimental Procedures
The bioceramic material with composition (wt%); 45 SiO2, 24.5 Na2O, 24.5 CaO, 6 P2O5 was prepared through the sol-gel process using bentonite clay as precursor. According to the extraction procedure reported by Essien et al. [26], a mixture of bentonite clay (13.33 g) in 1 M NaOH (250.00 ml) was heated at 120˚C for 2 hours under reflux in a round bottom flask. The filtrate obtained was slowly evaporated to dryness at 130˚C to obtain sodium metasilicate (Na2SiO3). In a typical gel-derived procedure for preparing Bioglass® 45S5 [15], Na2SiO3 (9.00 g) was dissolved in deionised water (15.00 ml) to give a solution that was then added slowly to 1 M HNO3 (15.50 ml) in a beaker furnished with a magnetic stirrer under ambient temperature for 1 hour to facilitate complete hydrolysis. Thereafter, P2O5 (0.59 g), NaNO3 (0.37 g) and Ca(NO3)2∙4H2O (10.16 g) were added respectively under constant stirring. Each of those three reagents was allowed 45 minutes to react. After complete addition, the mixture was further stirred for additional 1 h to afford a gel. The resulting gel was aged at room temperature for 5 days, dried at 60˚C, 200˚C, and 600˚C for 72, 40 and 5 hours respectively, followed by sintering at 800˚C for 3 hours and 1000˚C for 2 hours.
2.3. Methods of Characterization
Atomic absorption spectroscopy (AAS) Perkin Elmer A Analyst 200 was used to determine elemental composition of the finally sintered material. Further evaluation was by X-ray diffraction (XRD, X’pert PRO PANalytical) using CuKα radiation (0.154060) source operated at 40 kV and 40 mA. The diffraction patterns were obtained in the 2θ range from 5˚ - 120˚. The morphology of the material was investigated using scanning electron microscopy (SEM, EVO/MAIO). The sample was carboncoated and observed at an accelerating voltage of 10 kV. Fourier transform infrared (FTIR, Perkin Elmer Spectrum X), with the wavenumber range of 4000 - 400 cm−1 employing KBr pellets operating in a reflectance mode with a 4 cm−1 resolution was used to monitor the nature of bonds present in the glass network.
3. RESULTS AND DISCUSSION
3.1. Composition of the Porous BTC-Derived Bioglass Material
Bentonite clay of the type (OH)4Si8Al4O20∙nH2O shown in Figure 2(a) was reacted in strong basic medium to initially afford sodium metasilicate (reaction equation is shown elsewhere [26]) after evaporating the alkaline filtrate slowly to dryness at 120˚C. The waterglass obtained, Figure 2(b), from this reaction is hydrolysed under acidic conditions to silicic acid which undergoes condensation to disilicic acid and further reaction to a polycondensed hydrogel [26,27]. The elemental composition of the bioceramic material was adjusted to give a ratio similar to Bioglass® 45S5 by varying the amount of the following reagents; P2O5, NaNO3 and Ca(NO3)2∙4H2O.
The monolith obtained is shown in Figure 2(c). The weight percentage analysis by AAS of the monolith was confirmed as; (wt%) 45 SiO2, 24.5 Na2O, 24.5 CaO, 6 P2O5.
3.2. FTIR Characterisation of the Bioceramic Monolith Similar to Bioglass® 45S5
The FTIR spectrum of the BTC-derived bioglass monolith material is shown in Figure 3. It is characterised by a very broad OH absorption band centred around 3424 cm−1 and a weak absorbed water band at 1650 cm−1 [28]. Two other absorptions bands observed at 905 and 1011 cm−1 correspond to asymmetric Si-O-Si stretching vibration of silanol groups [29,30]. The peak centred at 565 cm−1 can be attributed to the P-O bond in crystalline phosphates [31]. Similarly, the weak peak at 1212 cm−1 is attributed to the characteristic vibration of P=O bonds. Furthermore, the broad band centred at 1440 cm−1 is diagnostic of the vibrations of non-bridging PO2 in  groups or
groups or  originated from air during the sol-gel fabrication [32]. The vibrational modes obtained for the bioceramic monolith compare with those of biomorphic 45S5 bioglass scaffold [32].
originated from air during the sol-gel fabrication [32]. The vibrational modes obtained for the bioceramic monolith compare with those of biomorphic 45S5 bioglass scaffold [32].
3.3. XRD Characterisation of the Bioceramic Monolith Similar to Bioglass® 45S5
XRD pattern of the BTC-derived bioceramic sintered at 1000˚C for 2 h is shown in Figure 4. It can be seen that the material contains both amorphous and crystalline phases. The amorphous phase is characterized by a wave-like tendency in the baseline and diffused peak bottoms caused by the short-range ordering of the silicate structure in the glass [33]. The major crystalline phase Na2Ca2Si3O9 has hexagonal system based on the angular
 (a)
(a) (b)
(b) (c)
(c)
Figure 2. (a) Bentonite clay; (b) Sodium metasilicate and (c) bioceramic monolith with Bioglass® 45S5 composition.
locations and the intensities of the sharp diffraction peaks which match those of the standard PDF #22-1455 [16,34]. The same diffraction pattern was reported by previous studies on sintered 45S5 bioactive glass [14-16,32,34].
There are two additional peaks observed at 2θ = 23.13˚ and 32.62˚ assigned to the (111) and (211) planes of orthorhombic NaCaPO4 crystal (PDF #29-1193) [29].
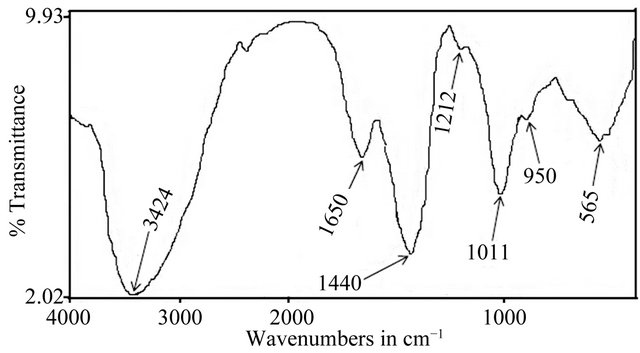
Figure 3. FTIR spectrum of the bioceramic monolith similar to Bioglass® 45S5.
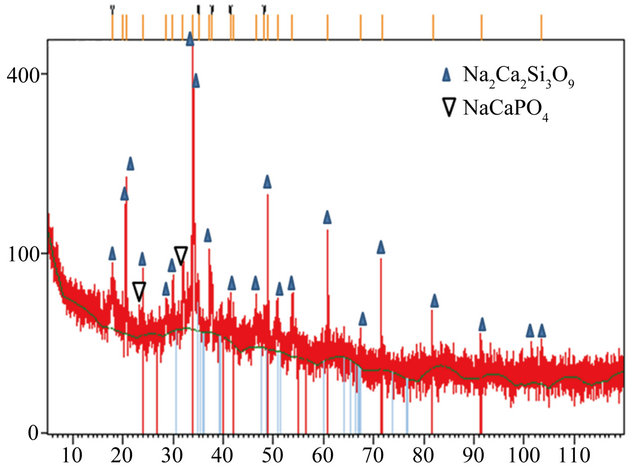
Figure 4. XRD pattern of the bioceramic monolith similar to Bioglass® 45S showing peaks for Na2Ca2Si3O9 and NaCaPO4.
3.4. Morphology of the Bioceramic Monolith Similar to Bioglass® 45S5
The porous nature of the BTC-derived bioceramic was investigated using SEM. Figure 5 shows that the material contains prominent macropores, micropores and pore struts. Most of the pores are open and interconnected, with a network of macropores linking the smaller micropores. Many authors agree that a minimum pore size of 100 µm is required for fast cell infiltration inside the scaffold material [27]. However, Figures 6(a)-(d) indicate the presence of seemingly widespread microporosity with an average size range of about 2 - 6 µm, and macropores that range between 20 - 70 µm. Similar morphologies have been reported for 45S5 bioactive glasses sintered up to 1000˚C [14,15].
Microporosity and the resultant large surface area are crucial to promote diffusion of fluids and nutrients by means of capillary in-growth [29]. The nature of the struts between pores as shown in Figures 6(c) and (d) is evidently thick and dense which is particularly required to enhance the scaffold mechanical strength.
4. CONCLUSION
Bioceramic monolith with elemental composition similar to Bioglass® 45S5 has been prepared by the sol-gel process employing bentonite clay as a cheap source of silica. Strong alkaline medium was used to initially obtain sodium metasilicate from the bentonite clay. The metasilicate was subjected to acidic hydrolysis to give the sol.

Figure 5. SEM micrograph of BTC-derived bioceramic showing macropores, micropores and pore struts.

Figure 6. SEM micrographs of the bioceramic at different magnifications. Arrows indicate pores in (a) and (b) and pore struts in (c) and (d).
Thereafter, elemental composition of the sol obtained was adjusted by addition of appropriate inorganic reagents to give the gel that underwent polycondensation reaction that gave a monolith after aging and drying. The monolith was subjected to phase transformation during sintering at elevated temperature to give a major crystal phase identified as Na2Ca2Si3O9 and a secondary crystal phase, orthorhombic NaCaPO4. XRD analysis indicated almost complete conversion of the amorphous phase to a new crystalline phase. Sintering also resulted in densification of the pore struts which is envisioned to enhance the mechanical property. FTIR analysis indicated characteristic absorption bands for Si-O-Si and silanol groups in the framework. SEM analysis however reveals that the pore sizes obtained are smaller than the ideal for bone ingrowth which is between 300 - 400 μm. Nevertheless, our results suggest that bentonite clay may serve as low cost raw material for preparing sodium containing bioactive glasses.
5. ACKNOWLEDGEMENTS
The authors are grateful to Bells University of Technology, Ota, Nigeria for providing funding and their laboratory for this work.
NOTES