1. Introduction
With the society and economic development, the problem of environmental pollution is outstanding day by day. Especially, the contamination of water due to color effluents coming from different industries, such as textiles, dyestuff, and paper, which are toxic and low-biodegradable, discharges into the aquatic systems and leads to the serious destroy of the survival environment as well as serious damage to human health [1] -[5] . Among these pollution, azo dyes, characterized by the presence of the N=N linkage, are the mostly and frequently used for colorization in textile industries [6] . Faced with the global environmental pollution, the process as efficient as possible in the azo dyes degradation has widely been pursued [7] . Several studies of physical, chemical and biological methods have been carried out. However, since their very stable and complex aromatic structures, the traditional treatment means used for azo dyes effluents are sometimes ineffective, non-destructive and merely transfer pollutants from water to another phase, resulting in secondary pollution, and even generating greater toxicity aromatic amine materials [8] - [10] . In recent years, the photocatalytic process on semiconducting materials has greatly been concerned because it provides a green way to directly degrade waste water pollutants [11] . As a result, numerous semiconductor photocatalytic materials, such as TiO2 [12] , SnO2 [13] , ZnO [14] , and BiVO4 [15] , have been developed and applied in photocatalytic degradation of organic pollutants due to their high efficiency, commercial availability and high chemical stability.
Perovskite oxides with the general formula of ABO3 in which A-site (with ionic radius of larger than 1.0 Å) is a rare-earth or an alkali-earth element and B-site (having ionic radius in the order of 0.6 - 0.8 Å) is typically a 3D transition metal become the hotspot of material science because of their stable structure and unique physical and chemical properties [16] . The ideal perovskite ABO3 has a cubic crystal structure, which is composed of a three-dimensional framework of eight corner-sharing BO6 octahedrons. Among the structure, BO6 octahedron is often considered as the basic cell and B-site cation locates in octahedral vacancy while A-site cation fills the twelve coordinate cavities formed by BO6 network [17] [18] . Due to their unique properties, such as various types of oxygen vacancy order, intrinsic oxygen reduction reaction activity, high conductivity and magnetic properties, perovskites have been used in solid oxide fuel cells, oxygen separation, membrane reactor for partial oxidation of methane to syngas and also used as catalysts for controlled partial hydrocarbon oxidation and photocatalysis [19] - [21] . Especially, their high performances in photocatalytic reactions, such as overall water splitting and photo degradation of organic pollutants, attracted much attention because of their narrower band gap (often less than 3.0 eV) which could be easily excited under visible light or ultraviolet light irradiation [22] . As a photocatalyst, perovskites, such as SrSnO3 [23] , SrTiO3 [24] and LaTiO3 [25] , have been extensively studied in the field of photo degradation of organic pollutants.
Double-perovskite oxides which named after perovskites ABO3 are a large family and have wide application fields. Among them, the oxygen deficient ordered double perovskite cobaltates with the general formula LnBaCo2O5+δ have drawn significant interest as potential IT-SOFC (Intermediate Temperature-Solid Oxide Full Cell) cathodes, chemical gas censors, magnetic and oxygen permeation materials because of their high electronic con- ductivity, oxygen transport properties, oxygen surface exchange coefficient and superior oxide ionic diffusivity [26] - [29] . The crystal structures of these oxides are formed by stacking sequence CoO2|LnOδ|CoO2|BaO|CoO2 along the c direction, and the oxygen vacancies are mainly localized into LnOδ layer. Transformation of a simple cubic perovskite with randomly occupied A-sites into a layered crystal with alternating lanthanide and alkali- earth planes reduces the strength of oxygen binding and provides disorder-free channels for ionic motion [30] . Since perovskite oxides possess high photocatalytic performances, as with close links to the perovskite compounds, double-perovskite oxides can be conjectured that they are a kind of potential photocatalytic material. Nevertheless, little work has been carried out to investigate their photocatalytic performances.
In this paper, the mainly research is focused on the preparation process and removal properties of organic pollutants CR from the wastewater using the double-perovskite oxides LnBaCo2O5+δ (Ln = Eu, Gd, and Sm).
2. Experimental
2.1. Materials Synthesis
All the chemical reagents used in the experiments were obtained from commercial sources as guaranteed-grade reagents and used without further purification and treatment.
LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders were prepared by a conventional solid-state reaction method. Ln2O3, BaCO3 and Co2O3 served as the starting materials for the necessary metal ions. The process flow chart is shown in Figure 1. Typically, Ln2O3, BaCO3 and Co2O3 powders were mixed in stoichiometric ratios and thoroughly ground in an agate mortar using ethanol as a solvent for 1 h. After the evaporation of ethanol, the mixed powders were calcined at 1000˚C for 6 h under the air atmosphere and then slowly cooled down to room temperature in the furnace.
2.2. Materials Characterization
The crystal structures of the synthesized materials were characterized by X-ray powder diffraction (XRD) using
![]()
Figure 1. Technological synthesis process of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders via solid-state reaction me- thod.
a Rigaku D/Max-3B instrument with copper target and Kα radiation (λ = 1.54056 Å). The accelerating voltage and applied current were 40 kV and 200 mA. The samples were scanned from 10˚ to 90˚ (2θ) in steps of 0.02˚.
2.3. Measurement of Photocatalytic Activity
CR (C32H22N6Na2O6S2, molecular weight: 696.67 g∙mol−1, from Guangzhou Reagent Corporation, China) was used as a model dye to evaluate the photocatalytic activity of the LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) samples. The degradation tests of CR were conducted in an aqueous solution at room temperature under ultraviolet light irradiation with the dye solution concentration of 25 or 50 mg∙L− 1 . In a typical degradation process, under ultraviolet light irradiation and stirring conditions, 20 mg of the synthesized samples were added into 50 mL CR aqueous solutions with an initial concentration of 25 or 50 mg∙L−1 in a quartz beaker, respectively. At a given time, 4 mL of the dispersion was continually extracted and subsequently centrifuged to separate LnBaCo2O5+δ powders and dye solutions at 4000 rev∙min-1 for 30 min. The reaction mixtures were irradiated by a Xe-lamp (30 W) with the wavelength range of 320 - 400 nm in which a peak wavelength of 365 nm for UV light and 400 - 800 nm for visible light, and magnetically stirred throughout the photocatalytic experiment under air. The initial concentration (C0 ) and the instant concentration (C ) of the aqueous solution of CR were determined with a UV-2401PC spectrophotometer at λmax = 498 nm . The degradation rate of CR dye was calculated as C/C0.
3. Results and Discussion
3.1. The Crystal Structures of the As-Synthesized Samples
Figure 2 shows the X-ray diffraction patterns of the oxides which prepared from the appropriate chemical precursors via calcination at 1000˚C for 6 h under an air atmosphere. The XRD patterns of samples with the nominal compositions of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) are represented at a single-phase double-perovskite without any impurity phase. All the diffraction peaks of LnBCO samples can be indexed well with orthorhombic structure EuBaCo2O5.52 (JCPDS No. 53-0136, a = 3.915 Å, b = 3.882 Å, c = 7.549 Å), GdBaCo2O5.42 (JCPDS No. 53-0135, a = 3.918 Å, b = 3.879 Å, c = 7.545 Å), SmBaCo2O5.54 (JCPDS No. 53-0133, a = 3.913 Å, b = 3.889 Å, c = 7.576 Å), space group Pmmm, and these findings are in agreement with previously published reports [31] - [33] .
3.2. The Photocatalytic Activity of the LnBCO Powders
The as-synthesized LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders with well crystal were used for photocatalytic tests under ultraviolet light irradiation. CR was used as a test contaminant since it has been extensively used as
![]()
Figure 2. X-ray diffraction analysis of the LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders.
an indicator for the photocatalytic activity. After photocatalysis experiments, we found that double-perovskite oxides LnBaCo2O5+δ could remove the CR effectively, which mean they have photocatalytic properties. Figure 3 shows the photodegradation rate of CR solutions with different initial concentration in the presence of samples. From Figure 3(a), it is easy to see that the degradation rate of CR solutions (C0 = 25 mg∙L−1) by EuBaCo2O5.52 is rapid at the first 16 h of the reaction time. Following the rapid degradation, the rate decreased and the decolorization efficiency was 92.6% for 24 h. The other two samples, GdBaCo2O5.42 and SmBaCo2O5.54, display similar photocatalytic degradation rates of CR comparing with EuBaCo2O5.52. In general, the catalytic activity is tightly related to the structure of the catalyst [34] . Double-perovskite oxides LnBaCo2O5+δ is a kind of distorted perovskite structure which is often caused by the excessive absence of O2− ion. The same structure can explain the phenomenon of similar photocatalytic property of EuBaCo2O5.52, GdBaCo2O5.42 and SmBaCo2O5.54. And likewise, as shown in Figure 3(b), because of the same reason, the three samples have similar photocatalytic degradation rates of a higher concentration of CR solutions (C0 = 50 mg∙L−1) except the time of photodegradation extended to 60 h.
To further clarify the photocatalytic performance and mechanism of CR, the continuous UV-Vis spectra of the centrifuged solution after catalytic reactions at the different intervals were used to record and in contrast to initial CR solution. Figure 4 displays the UV-Vis absorption spectra of CR solutions (C0 = 25 or 50 mg∙L−1) before and after treatment with as-synthesized LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders. From the UV-vis absorbance spectrum of CR initial solution, we can see three absorbance peaks at 241, 342 and 498 nm, respectively. The peaks at 241 nm and 342 nm are attributed to benzene ring and naphthalene ring structures, while the peak at 498 nm is attributed to the azo bonds of CR molecule [35] . The characteristic absorption peaks of CR at 342 nm and 498 nm were chosen as the parameter that was monitored. As shown in Figure 4, in the presence of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders, all absorption peaks went down dramatically along with increasing degradation time and correspondingly the solution became decolorized from red to near colorless. The amplitude of the characteristic peaks continuously decreased with respect to the reaction time and finally to disappear in Figure 4 suggests that the azo bonds and the naphthyl rings were destroyed to form phenyl derivatives. UV light illumination of the aqueous CR solution in the presence of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders causes the absorption bands of the CR dye in the visible region to disappear basically, which indicates that the dye is degraded completely. When LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders used to degrade CR aqueous solutions, the color of solution was bleached and the absorption of solution at 498 nm was close to zero. In accordance with changes of the absorbance intensity at 498 nm, the decolorization efficiency of 25 mg∙L−1 CR solution in the presence of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders after 3 h degradation reaction was
![]() (a)
(a)![]() (b)
(b)
Figure 3. The degradation rate of CR solutions with different initial concentration ((a) 25 mg∙L-1 and (b) 50 mg∙L-1) under ultraviolet light irradiation at room temperature in the presence of EuBaCo2O5.52, GdBaCo2O5.42 and SmBaCo2O5.54, respectively.
achieved about 47%. Complete decolorization of CR solution (C0 = 25 mg∙L−1) was achieved within 26 h. These results are in agreement with the analysis shown in inset. It is clearly seen that the cardinal red color of starting solution gradually disappears along with increasing the degradation time, which also depicts the degradation process of CR.
The possible catalytic mechanism of LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders is proposed like that of the perovskite oxides. We suppose the photocatalytic property of double-perovskite is derived from the BO6 octahedron. In LnBaCo2O5+δ, 2p-orbit of O2? constitutes the valence band, while the 3d-orbit of Co3+ constitutes the conduction band. Under the ultraviolet light irradiation, the valence bands electrons of LnBaCo2O5+δ were excited and transferred into conduction bands, generating highly reactivity electron-hole pairs which can induce oxidation of organic molecule, by leaving holes (h+) in the valence bands. The hole (h+) is a strong oxidant on the surface of LnBaCo2O5+δ and could oxidize the adsorbed water molecules or hydroxyl ions into ・OH radicals (H2O or OH? + h+ → ・OH ) which is highly radicals to oxidize or degrade the adjacent CR dye molecules. At the same time, the adsorbed oxygen reacted with electrons to form  superoxide ions (O2 + e? →
superoxide ions (O2 + e? →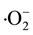 ) which could decompose the organic molecules effectively [23] [36] .
) which could decompose the organic molecules effectively [23] [36] .
![]()
Figure 4. UV-Vis absorption spectra of CR solutions (25 or 50 mg∙L-1) before and after treatment with as-synthesized LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders at different time intervals under ultraviolet light irradiation at room temperature: (a) EuBaCo2O5.52, CR (25 mg∙L-1); (b) EuBaCo2O5.52, CR (50 mg∙L-1); (c) GdBaCo2O5.42, CR (25 mg∙L-1); (d) GdBaCo2O5.42, CR (50 mg∙L-1); (e) SmBaCo2O5.54, CR (25 mg L-1); (f) SmBaCo2O5.54, CR (50 mg∙L-1). The insets are the photographs of CR solutions (25 or 50 mg∙L-1) before and after treatment with as-synthesized LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders at different time intervals, respectively.
4. Conclusion
Double-perovskite oxides, LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) microparticles were successfully prepared by a conventional solid-state reaction method. The samples were characterized by X-ray diffraction (XRD), showing that the resulting particles were highly crystalline LnBCO particles. The photocatalytic activity of the LnBaCo2O5+δ (Ln = Eu, Gd, and Sm) powders was investigated by the degradation of CR, and the results revealed that LnBCO had a certain photocatalytic activity, which indicated that they could be a promising photocatalyst for the degradation of organic molecules.
Acknowledgements
This work was supported by National Natural Science Foundation of China (Grant No.51262029), the Key Project of the Department of Education of Yunnan Province (ZD2013006), Program for Excellent Young Talents, Yunnan University (XT412003), Yunnan University Graduate Program for Research and Innovation, and the Department of Science and Technology of Yunnan Province via the Key Project for the Science and Technology (Grant No.2011FA001).
NOTES
*Corresponding author.