Searching Clue of the Relationship between the Alteration of Bioactive Ingredients and the Herbal “Property” Transformation from Raw Rehmanniae Radix (Sheng-Di-Huang) to Steam-Heating-Processed Rehmanniae Radix (Shu-Di-Huang) by Chromatographic Fingerprint Analysis ()
Run time: 55 minutes Compare the chromatogram of sample solution against the chromatogram of reference solution.
3. Results and Discussion
3.1. HPTLC Fingerprint Analysis
Characteristics HPTLC fingerprint The HPTLC fluorescence image of raw dried Rhamannia roots (Sheng-Di-Huang) developed four yellow fluorescent bands of saccharides: stachyose (Rf value ac. 0.06), raffinose (Rf value ac. 0.14), sucrose (Rf value ac. 0.26), D-fructose (Rf value ac. 0.35) and two grayish-blue fluorescent bands of iridoids (rehmannioside D (Rf value ac.0.18), catalpol (Rf value ac.0.52). The relative fluorescence abundance of the components in the image can be compared visually (Table 1).
The digital scanning profiles of the HPTLC images (Figure 1) expressed more clearly the intact view of the fingerprint of Sheng-Di-Huang. It can serve as the common pattern of the raw material (Figure 2). As a typical Sheng-Di-Huang, among saccharides, strachyose is the strongest fluorescence band, raffinose and sucrose gave stronger or weaker fluorescence bands, fructose is the weakest in different samples; As iridoids, catalpol were obviously detected as grey-blue fluorescence bands, but the content (fluorescence abundance) fluctuated widely

Figure 1. HPTL Cimage of Di-Huang (Rehmanniae Radix). (S1)-Stachyose; (S2)-Raffinose; (S3)-Rehmannioside; (S4)-Surcose; (S5)-D-Fructose; (S6)- Catalpol; (1). Reference crude drug; (2)-(6) Commercial samples; (S1~6) mixed chemical reference substances.

Figure 2. HPTLC images and digital scanning profiles of mixed CRS and HRS of Rehmannia root. Peaks: (1) stachyose; (2) raffinose; (3) rhemannioside D; (4) sucrose; (5) fructose; (6) catalpol, (?) unknown yetsaccharide.

Table 1. Approximate comparison of the fluorescence bands abundance of the saccharides and iridoids in the HPTLC image of Sheng-Di-Huang and Shu-Di-Huang.
ª: glucose overlapped with fructose.
in different commercial samples (The literature reported catalpol is the highest content among the iridoids glycosides [11] ). Briefly, the intact image and the scanning profile (number of bands, location and order, fluorescence/visible color, strength of the fluorescence abundance, peak height/area ratio between bands) constructed the unique fingerprint pattern of Sheng-Di-Huang.The digital scanning profiles of the HPTLC images (Figure 1) provided more clear intact view of the fingerprint. It can serve as the common pattern of Sheng-Di-Huang (Figure 2).
With regard to the HPTLC image pattern of Shu-Di-Huang, comparison with of Sheng-Di-Huang, the prominent feature is iridoids represented by catalpol were decomposed seriously, even radically disappeared. Secondly, the oligosaccharides were hydrolyzed at different degrees, consequently sucrose, even fructose increased accordingly (Table 1). Therefore the HTPLC image of Shu-Di-Huang showed a rather different visage from ShengDi-Huang. Glucose almost dehydrated to 5-hydrxoy-methyl furfural (5-HMF) (vide infra).
Dynamic alteration of the active ingredients of the root tuber of Rehmannia subjected to the steaming process For the sake of investigation of the dynamic alteration of the compounds in Di-Huang, a quantity of “ShengDi-Huang” was steamed in a small bamboo steamer placed in a wok filled with boiling water, heated for 0, 2, 4 and 8 hours; and then a given quantity was taken out as samples, dried under reduced pressure at 60˚, powdered, then prepared the sample solutions as described in section 2.2. for HPTLC analysis. The experiment demonstrated the steam-heating tolerance test of oligosaccharides and catalpol. The results indicated that stachyose, raffinose and sucrose in the roots were at rather stable till 4 hours. After that, those all gradually hydrolyzed drastically in different extent, and the D-fructose proportional increased, but stachyose was relatively rather stable against steam-heating (Figure 2). It means that the majority of the oligosaccharides were hydrolyzed to monosaccharide—D-fructose and glucose, only stachyose still exist. Catalpol—the main iridoid glycoside seriously decreased, even radically disappeared (Figure 3).
On the other hand, the chain-structured monosaccharide like glucose would be decomposed to be 5-hydroxymethylfurfural (5-HMF). The increase trend of 5-HMF along with the steaming period was occurred simultaneously (Figure 4).
Application of fingerprint to the commercial samples analysis Generally the commercial samples of Sheng Di-Huang and Shu-Di-Huang collected from various markets, the

Figure 3. Comparison of peak area of several active components of Di-Huang (Rehmanniae Radix) steamed in persistently prolonged heating.
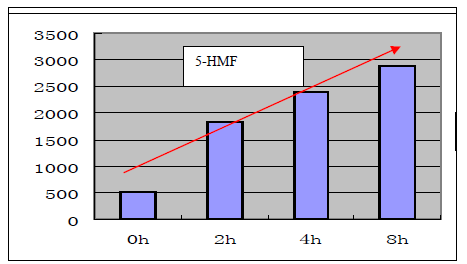
Figure 4. Peak area of 5-hydroxy-methylfurfural (5-MHF) inDi-Huang which was generated from decomposition of glucose when steamed in persistently prolonged heating.
labeled names rely solely on the claims of the suppliers. From the chromatographic fingerprints it is apparent that the ingredients in various samples exhibited some fluctuation which caused by variations in determining methods, sample habitats and uncontrolled steam-heating process [12] [13] .
Based on the extent of decomposition of the ingredients, there are four grades of the patterns in the collected commercial samples as shown in the HPTLC fingerprints (images combined with the corresponding scanning profiles) (Figure 5). It is noteworthy that the most important tetra-oligosaccharide—stachyose [14] seems relatively more stable than the other oligosaccharides (Figure 5 and Figure 6).
The project plot of Principle Component Analysis (PCA) of “Di-Huang” disclosed that while some of the commercial samples of “Sheng-Di-Huang” were closely distributed around the common pattern, (Grade I), others spread a distance from the common pattern, located at the upper part (Grade II) which have the typical character of of “Sheng-Di-Huang” common pattern, but catalpol was hard to be detected. Grade III and Grade IV those in the right side belonging to “Shu-Du-Huang”. An exceptional example of Shu-Di-Huang (21#) showed only fructose. (The intact root tuber is of “as black as Chinese lacquer” (e.g., #21 sample) and always being considered as superior entity in the description of ancient herbal compendia). Obviously in fact such sample suffered from over-steaming (Figures 5-7).
3.2. HPLC Fingerprint Analysis
HPLC fingerprint of Di-Huang For analysis of the phenylethanol derivatives in Di-Huang, the HPLC-DAD experiment was carried out, the

Figure 5. The HPTLC images of “Di-Huang” are divided four grades. (The figure below is the inverted color image from the upper fluorescence figure for more easy comparison), Track (1) Chemical reference substances: (1) stachyose, (2) raffinose, (3) rehmannioside D, (4) sucrose, (5) fructose, (6) catalpol Track (2) authentic sample of “Sheng-Di-Huang”; Track (3) commercial sample of “Sheng-Di-Huang” Track (4) similar appearance of the crude drug with the authentic sample (2) pattern, but catalpol exceptionally disappeared and the rehmanioside D was very weak. (cf. Figure 7). Track (5)-(6) the crude drugs appearances and the HPTLC pattern of the commercial samples were up to the description of “Shu-Di-Huang” in Chinese Pharmacopoeia, but the extent of hydrolysis of the saccharides differ, the monosaccharide-fructose increased drastically. Note that the stachyose (band 1) was relatively more stable than the others. Track (7) an over-steamingprocessed sample of “Shu-Di-Huang”, almost all the major ingredients disappeared or only residual amounts remained, but fructose being abundant. The blue stars marked in the invert color images (the figure below) showed some weak chemicals which are almost hard to be recognized under deep color background in the original fluorescence image.

Figure 6. PCA projection plot of HPTLC fingerprint of Di-Huang. The Grade I and Grade II belong to “Sheng-Di-Huang”, but the samples in Grade II, catapol was hard to be detected. Grade III and IV expressed the feature of “Shu-Di-Huang”, but the samples in Grade IV contain minor amount of catalpol. The dynamic change of the oligosaccharides accompanying the steam processing caused some samples (“Shu-Di-Huang”) distance from the common pattern. The longer the steam-heating time, the more severe the hydrolysis of raffinose and sucrose transforming them into fructose and glucose will occur. Stachyose was rather stable than the other oligosaccharides. (cf. Figure 5). Sample #21(“SDH”) showed only fructose (+ glucose) dominated in the fingerprint due to over-steam-heating led almost all oligosaccharides hydrolyzed to monosaccharide.
detection wavelength set at UV 334 nm to meet the maximum absorbance of this kind of components. The HPLC common pattern of “Sheng-Di-Huang” consisted of 11 characteristic peaks including acteoside and other phenylethanols (Figure 8).
Applied Fingerprint to commercial sample analysis Similarity of commercial samples has been calculated based on correlation coefficient for comparison with the common pattern (CP) (see Figure 9).
Since the concentration, distribution and proportion between the active components differ from reference crude drug, some samples of relative lower similarity (below 0.85) compared with the common pattern. Samples q#14 and #30 are two examples with unusual profiles individually. The supernormal strong peak of acetoside in HPLC profile of sample #14 also led to lower similarity (correlative coefficient 0.83) and oppositely a severe decline peak of acetoside as well as the peak (3) and (4) in the HPLC profile of sample #30 caused the lowest similarity of 0.24 (Figure 10 and Figure 11).
Comparison of HPLC fingerprints between Sheng-Di-Huang and Shu-Di-Huang In HPLC fingerprint of the phenylethanols derivatives of Shu-Di-Huang are also decomposed in some extent by comparison with “Sheng Di-Huang”, and increased some of the higher polarity compounds in the region of ca. Rt 4 min to 14 min (Figure 12).
4. Discussion
Combining the HPTLC fingerprint (fluorescence images plus the corresponding digital scanning profiles) and

Figure 7. The changes of the HPTLC fingerprints of various commercial DiHuang sample. (1)-Stachyose; (2)-Raffinose; (3)-Rehmannioside D; (4)-Surcose; (5)-D-Fructose; (6)-Catalpol.
the HPLC-DAD fingerprints of “Di-Huang” constructs an integrative view on the characteristics distributions of ingredients in “Sheng-Di-Huang” and “Shu-Di-Huang”. The HPTLC fingerprint showed a whole range of detectable iridoids and saccharides. HPLC gave the profile of phenylethanol derivatives. The fingerprints provided useful information to distinguish Di-Huang by means of dual chromatographic analysis. It was revealed that all the bioactive ingredients in Di-Huang are steam-heating-labile. The fingerprint of Sheng-Di-Huang consists of catalpol, rehmannioside D, stachyose, raffinose, sucrose, and fructose as well as some minor unknown components. But those chemical ingredients led to decompose during the steaming process. The loss of catapol and other iridoids and varying degrees of decomposition of saccharides exhibited in the fingerprints of Shu-DiHuang. The dynamic change of the saccharides in HPTLC fingerprint can possibly be traced to the extent of the

Figure 8. HPLC fingerprint common pattern of dried raw Rehmmaniae radix (Sheng-Di-Huang).

Figure 9. Similarity results of HPLC fingerprint of all samples of Di-Huang.
steaming process. It is noteworthy that stachyose seemed rather stable than the other oligosaccharides as expressed in the fluorescence images of various samples of “Shu-Di-Huang”.
Referring to phenylethanol derivatives, acteoside which dominated in the HPLC fingerprint of “Sheng-DiHuang” also decline gradually in Shu-Di-Huang as the steaming process went on.
In a word, “Shu-Di-Huang” cannot preserve the intact appearance of the fingerprint like that of Sheng-DiHuang, either HPTLC (iridoids and oligosaccharides) or HPLC (phenylethanol derivatives) due to instability of the said bioactive metabolites. On the other hand, an ensuing rising of 5-HMF derived from glucose was also a complementary feature of Shu-Di-Huang (Figure 4). Recent studies claimed that 5-HMF is likely also an important bioactive component which exists in many herbal drugs with the function of antioxidant and nourishing kidney Yin deficiency [15] . So its influence to the drug property is also worth considering.
As iridoids and hydrolysable oligosaccharides are unstable, it brought up the possibility of developing a line

Figure 10. Comparison of HPLC profiles of commercial sample 14# with common pattern of “Sheng-Di-Huang”. the content of acteoside is much higher than that in the common pattern.

Figure 11. Comparison of HPLC profiles of sample 30# and the common pattern. The acteoside and other phenylethanol compounds severely declined in sample 30#,the appearance of which looked as Sheng-Di-Huang.

Figure 12. Comparison of HPLC profiles of Sheng-Di-Huang and Shu-Di-Huang.
to divide Di-Huang from “Sheng-” (raw, dried) to “Shu-” (processed, steam-heated). The dynamic alteration of the bioactive ingredients provided some clue on the correlation with the herbal drug’s “property” transformation. It might presume from the fingerprint that the iridoids (catalpol is the highest content [8] ), in particular, with “cold” property in Sheng-Di-Huang to exert the principle role for “cooling the hot of the blood”, etc.; oligosaccharides, fairly stable stachyose with “warmer” property in Shu-Di-Huang to exert the function of nourishing “yin” of the “kidney”, etc. It cannot certainly be neglected that the holistic role with the other chemical components, i.e., phenylethanol glycosides, polysaccharides, etc.; in addition, the 5-HMF derived from dehydration of glucose and fructose during processing has also its contribution to the property transformation in Shu-Di-Huang. But the obvious different distributions of the detectable bioactive constituents in the two kinds of Di-Huang demonstrated in the chemical fingerprints are inferable that the properties of the both Di-Huang have their own individual focus and cannot be considered equivalent based on bioactivity. Modern pharmacological studies have also disclosed the various bioactive functions of different constituents in Di-Huang [15] [21] . The analysis result confirmed that too long steam-heating for getting the root tuber black as lacquer recorded in the historic herbal books cannot be considered the effective bioactive ingredients composition retained. Hence, standardizing the process procedure is necessary. Supposedly, it would be feasible to steady the quality of Shu-Di-Huang with the measures like cutting the root tuber into small pieces, improving the facilities, fix the time of steaming, enhancing the in-process quality control. Further in-depth investigation on the causal relationship between the chemical fingerprints and the traditional recorded Chinese medicine uses in a holistic way can be expected in the future.
5. Conclusion
The inconsistent chemical profiles of Shu-Di-Huang demonstrate that traditional processing methods can sometimes be of questionable value. Traditionally, nine cycles of “steam-heating and drying” process would be carried out turning the color to be as black as lacquer to obtain “the best quality” of Shu-Di-Huang according to traditional descriptions in the ancient Compendium Materia Medica. However there was no standardized procedure to regulate the operation and no real criteria for evaluation in spite of the request of the root tuber’s color. The results of chromatographic fingerprint analysis confirmed that the uncertainty of the total steam-heating procedure makes the quality of the final product inconsistent, even some over-steamed Shu-Di-Huang caused a loss of almost all bioactive components and only fructose remained. It is appealed to the manufacturer the significance for defining the appropriate standard for both Sheng-Di-Huang and Shu-Di-Huang. Referring to the pattern of chromatographic fingerprints, the iridoids should be preserved as more as possible in “Sheng-Di-Huang”, and the oligosaccharides, stachyose in particular, should be kept as consistent as possible in “Shu-Di-Huang”, the increment of 5-HMF caused by dehydration of glucose in processing period should also be measured as it has its bioactive contribution.
Acknowledgements
The corresponding author thanks Professor Du Hengqing for her help in obtaining authentic samples of Di-Huang.
NOTES
*Corresponding author.