1. INTRODUCTION
Flowering has been extensively studied in the model plant, Arabidopsis; where it is regulated by at least four parallel pathways, i.e., long day, vernalization, GA dependent, and the autonomous pathway. These pathways converge at a single point and switch on the expression of the floral meristem identity genes such as LEAFY (LFY), AP1 (APETALA1) and CAULIFLOWER (CAL) as described by [1-4]. Among the meristem identity genes, LFY is the first gene to be expressed preceding flower formation and the mutated form has the most drastic effects on the floral transition [5]. After turning on the meristem identity switch, the LFY gene has a second role as a transcription factor in the activation of the floral homeotic genes that specify the identity of organs in the flower [6,7]. Despite its pivotal role in the development of Arabidopsis thaliana, the molecular events downstream of LFY activation (e.g. signaling and regulatory steps, the identity of the genes regulated by LFY) are poorly understood. Among the few direct target genes of LFY that are currently known are APETALA1 (AP1), AGAMOUS (AG), and APETALA3 (AP3) [2,8-10].
The expression of LFY gene was reported to increase with the age of the plant till it reached a threshold level which is required for transition from vegetative to the reproductive phase [11]. Overexpression of the LFY gene has been found to accelerate flowering in a variety of plants like, aspen and tobacco [12], rice [13], poplar [14] and Citrus [15]. This ability to accelerate flowering has generated considerable interest in its role in transition from vegetative to the reproductive phase and its potential for modulating flowering in agriculturally important crops.
The LFY gene has been cloned and its function has been studied from a few species viz. Antirrhinum [16], tobacco [17], Eucalyptus [18], Pinus [19], rice [20], tomato [21], poplar [14], violet cress [22], apple [23], grapevine [24], papaya [25], rubber [26] and cedar [27].
In the present investigation Brassica juncea was selected as the experimental interest as it is an important oilseed crop of India. It is an amphidiploid (2n = 36, genome AABB) species raised by the hybridization of B. rapa (2n = 20, genome AA) and B. nigra (2n = 16, genome BB). B. juncea along with B. carinata provides 12% of the world wide edible vegetable oil supplies. When compared to other edible oils, the rapeseed/mustard oil has the lowest amount of harmful saturated fatty acids. It also contains adequate amounts of the two essential fatty acids, linoleic and linolenic acid, which are not present in many of the other edible oils. Our earlier studies showed accelerated flowering in transgenic B. juncea by overexpressing the LFY cDNA from A. thaliana [28]. In the present study with the future aim of expressing homologous LFY in B. juncea, we have isolated LFY from B. juncea and studied its temporal and spatial expression in B. juncea in different cultivars with differences in flowering time.
2. MATERIALS AND METHODS
2.1. Plant Material
Brassica juncea cv. Varuna, RNBL, TN-1, PNMB seeds were obtained from I.A.R.I, Pusa, New Delhi, India. The TN-1 is an early flowering cultivar while PNMB and RNBL are late flowering cultivars.
2.2. Cloning of LFY cDNA
Approximately 1 mg of total RNA isolated from floral buds of Brassica juncea was denatured at 65˚C for 10 min and reverse transcribed using MuMLV reverse transcriptase at 42˚C for 1 hr. An aliquot of the reverse transcribed cDNA formed was used for PCR with LEAFY cDNA specific primers. A mixture of Pfu and Taq Polymerase in the ratio of 1:3 was used for the amplification. A band of ~1.3 kb was obtained. The amplicon obtained was cloned in pGEMT-Easy vector, transformed in E. coli and the positive colonies identified by colony PCR. The positive clone was sequenced using automatic sequencer at DNA sequencing facility, Department of Biochemistry, University of Delhi, South Campus, New Delhi, India.
2.3. Analysis of LFY Expression Profile
RNA was isolated from freshly harvested tissues (floral buds, leaves, roots, stem, silique and shoot apices) of B. juncea according to a modified protocol of Chomozynski and Sacchi [29]. Total RNA was fractionated on 1.5% formaldehyde denaturing agarose gel according to Sambrook et al. [30] and transferred to nitrocellulose membrane in the presence of 20X SSC (3.0 M NaCl, 0.30 M sodium citrate, pH 7.0) by capillary transfer for 14 - 18 h. The blot was crosslinked by uv irradiation at 254 nm for 2 min. Hybridization with the radiolabeled BjLFY cDNA was carried out at 58 C in buffer containing 0.5 M sodium phosphate buffer, pH 7.2, 1 mM EDTA and 7% SDS for 16018 h. The membrane was washed twice in 1X SSC containing 0.1% SDS for 15 min at 58 C and kept in phosphoimager plates. The signals were detected by phosphoimager (FLA-500, Japan). Equal loading and proper transfer of RNA was monitored by methylene blue staining of the nylon membrane.
3. RESULTS AND DISCUSSION
3.1. Cloning and in Situ Characterization of LFY cDNA from B. Juncea cv. Varuna
A cDNA (DQ471932) encoding for LEAFY protein was cloned from B. juncea cv. Varuna. The deduced polypeptide comprised of 420 amino acids with predicted molecular weight 47 kDa and pI 6.48. The putative protein encoded by BjLFY in this study, shares a number of sequence motifs with a LFY/FLO like proline rich region near the amino terminus and a highly acidic region in the centre of the protein which are thought to be characteristic of plant transcription factors. These were present in the variable regions (low similarity) of the FLO/LFY proteins. The B. juncea LFY protein was evolutionary more closely related to the LFY proteins from A. thaliana and B. oleracea as was expected.
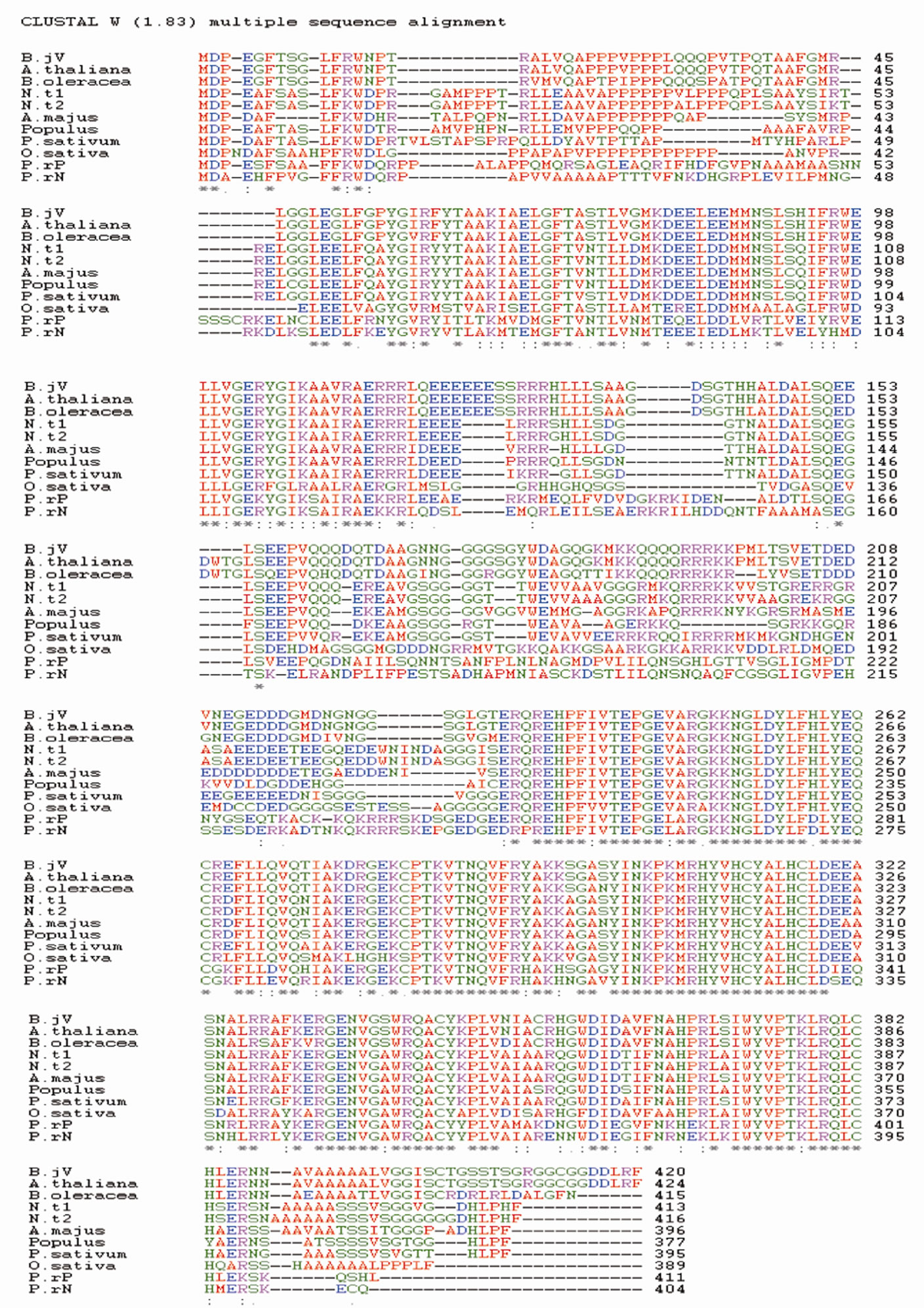
An alignment of the predicted amino acid sequence with the reported sequences revealed that BjLEAFY-V shares 99% identity with Arabidopsis thaliana LFY protein (AtLFY) and 86% identity with Brassica oleracea LFY protein (BOFH) at the amino acid level. Between BjLFY-P and BjLFY-V there was 88% identity. BjLFY-V also had the typical FLO/LFY regionsthe proline-rich region near the amino terminus and an acidic central region present within the encoded amino acid sequence. Sequence comparison of FLO/LFY-like proteins (accession numbers in parentheses) PrFLL from Pinus radiata (U92008); NLY from Pinus radiata (U76757); BOFH from Brassica oleracea (Z18362); LEAFY from Arabidopsis thaliana (M91208); NFL1 and NFL2 from Nicotiana tabacum (U16172 and U16174, respectively); PEAFLO from Pisum sativum (AF010190); FLO from Antirrhinum majus (M55525); PtFL from Populus balsamifera (U93196); and RFL from Oryza sativa (AB005620) was done with the Brassica juncea LFY amino acid sequence using ClustalW 1.83 (Figure 1(a)).
The amino acid sequence from 115 - 132 of BjLEAFYV correspond to the bipartite nuclear targeting sequences. The tools at Expasy could also identify four sites, 126 - 128; 229 - 331; 339 - 341and 408 - 410 in BjLFY-V. Besides these two caesin kinase II phosphorylation sites at 201 - 204 and 205 - 208, three sites at 150 - 153, 201 - 204 and 205 - 208 in BjLEAFY-V are also predicted to be present. SUMOplot™ predicted that the region 79 - 82 (MKDE) has a high probability to be engaged in SUMO attachment. Myristoylation sites were also predicted to be present in the proteins.
Dendrogram drawn by comparing the amino acid sequences of LFY proteins from Brassica oleracea, Arabidopsis thaliana, Nicotiana tabacum, Pisum sativum, Antirrhinum majus, Oryza sativa and Pinus radiata using the software Genebee (Figure 1(b)). As expected this analysis showed a close relationship between LFY proteins from B. juncea, B. oleracea and A. thaliana.
Southern blot analysis of B. juncea genomic DNA using the BjLFY-V cDNA as a probe gave multiple bands (Figure 1(c)). B. juncea is an amphidiploid species derived from B. rapa and B. nigra. The multiple signals obtained in B. juncea may probably be due to the presence of multiple copies of LFY in B. juncea genome inherited from each progenitor species or through the presence of these sites within the introns of LFY gene. In most plants for which LFY homologues have been determined, these LFY-homologous genes had a single copy in each genome (A. thaliana, A. majus, pea, tomato, petunia and poplar) [6,14,16,21,31,32]. In apple genome there are two homologous copies of LFY genes (AFL1
 (a)
(a)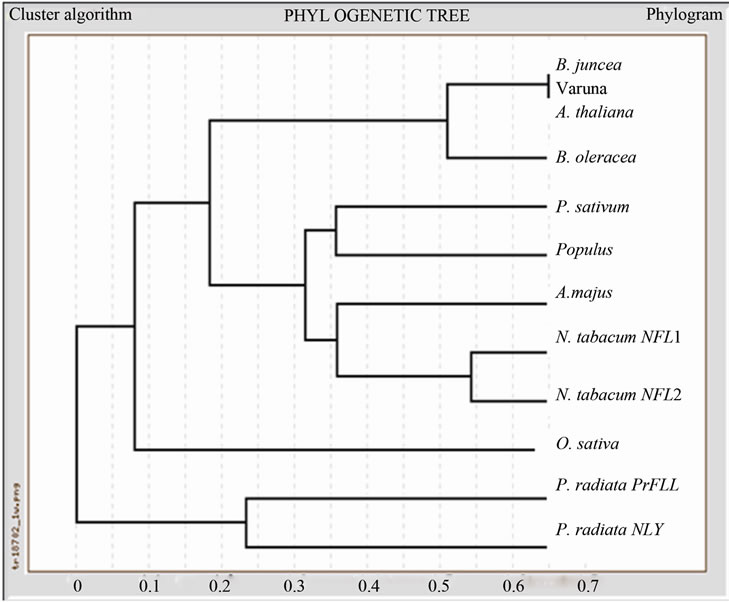 (b)
(b) (c)
(c)
Figure 1. (a) Multiple sequence alignment of BjLFY-Varuna. ClustalW was used for the multiple sequence alignment. “*” means that the residues or nucleotides in that column are identical in all sequences in the alignment, “:” means that conserved substitutions have been observed and “.” means that semi-conserved substitutions are observed; (b) Dendrogram of BjLFY-Varuna protein obtained using Genebee software; (c) Southern hybridization analysis of Brassica juncea cv. Varuna genome. Genomic DNA (20 μg) from Brassica juncea was digested with Bam HI (B), Eco RI (E), Hind III (H), Nco I (N), Pst I (P) or Sal I (S) at 37˚C overnight. The digested DNA was separated on 1% agarose gel, blotted on nylon membrane and hybridized with a probe prepared from a BjLFY cDNA. M indicates λ DNA EcoR I/digest. The numbers at right indicate molecular size (kilo basepairs).
and AFL2) [23] and so also in tobacco (NFL1 and NFL2) [17]. Eucalyptus has three homologous genes in the genome, but two of them were found not to be expressed and had stop codons in their coding region [18]. P. radiata has two homologous genes, NEEDLY and PRFLL which are expressed during vegetative development and in male cones during reproductive development respectively [19,33].
3.2. Spatial and Temporal Expression of BjLFY during B. juncea Development
The expression of LFY gene was studied in different organs, viz. adult leaves, root, stem, silique and bracts of B. juncea. Northern blot analysis revealed a very high expression of the LFY gene in mature leaves, followed by stems and bracts. There was very faint expression in the silique and no expression in the roots of the plants (Figure 2). The LFY gene expression was detected in all the four floral organs although there were differences in the level of expression. This is similar to the expression pattern observed in A. thaliana and A. majus [6,16]. The expression of the LFY gene in the floral organs of B. juncea reflects their role in establishing floral organ identity like the other FLO/LFY genes. Unlike BjLFY its homologue from other plants (BOFH gene from B. oleracea, ELF gene from Eucalyptus, AFL1 gene from apple and NFL genes from tobacco) have not been reported to express in leaf and stem [17,18,23,34].
As we observed considerable expression in leaves of B. juncea we decided to study the changes in LFY expression in leaves at different developmental stages. The LFY transcript could be detected very early in the cotyledonary leaves within one week after germination, after which it decreased during vegetative growth in the next two weeks. When the plant was ca. 26 - 34 days old

Figure 2. Expression of the LFY gene in different organs in wild type plants of B. juncea cv. Varuna.
with no visual signs of flowering, there was a sudden increase of LFY transcript, which was sustained till the appearance of the floral buds. The LFY transcript level decreased on initiation of flower development. Therefore, the kinetics of the LFY gene expression in B. juncea cv. Varuna suggested that there is an increase in level of LFY transcript during the transition from vegetative to the reproductive phase (Figure 3(a)). Most species for which expression of LFY genes has been investigated have simple leaves and where LFY expression occurs in leaves, it is low and restricted to early phases of leaf development (A. thaliana; [35], N. tabacum; [17], Impatiens species; [36]). An interesting observation by Lee et al. (1997) who reported the formation of lobed leaves in transgenic Arabidopsis plants overexpressing UNUSUAL FLORAL ORGANS (UFO) gene and this phenotype required the presence of functional LFY gene [37]. Therefore, LFY could indirectly be involved in controlling the morphology of simple leaves. In the present investigation, the LFY transcript in the leaves of B. juncea was abundant during the initial one week of growth and decreased during the vegetative growth. It showed an increase during the transition phase (~26-34-day-old) from the vegetative to the reproductive phase and decreased slightly when re-productive development started. The modulation of LFY gene expression during the vegetative to reproductive development in B. juncea suggests that it has an important role to play. An in-depth study is needed to support this.
The LFY expression was also studied in shoot apices of different cultivars at different time period starting from 10-day-old seedlings to 50-day-old adult plants. The LFY gene expression was detected very early in the shoot apices of B. juncea during the vegetative phase. The level of the transcript was more abundant in the early flowering cultivar, TN-1 followed by Varuna. The expression in PNMB and RNBL cultivars which are late flowering was very less in the early stages of growth (10 - 24 days after germination) but subsequently increased with age (Figure 3(b)). The appearance of the LFY transcript much earlier and at higher levels in early flowering cultivates indicate that the threshold level required for floral transition is attained faster in these as compared to the late flowering ones. Though the LFY and FLO genes are expressed in the floral apices, the NFL transcript could be detected in the vegetative apices also [17].
Although, the expression studies of LFY gene of B. juncea showed that it is expressed throughout the life cycle of the plant, there is a variation in the amount of transcript at different developmental stages and in different organs. Despite the differences in expression pattern, these expression studies indicate that BjLFY could be involved in regulating floral meristem identity and
 (a)
(a) (b)
(b)
Figure 3. (a) Northern blot of total RNA isolated from leaves of B. juncea cv.Varuna at different developmental stages; (b) Northern blot of total RNA isolated from shoot apices at different stages of development in various cultivars of B. juncea varying in flowering time.
flowering time as in LFY/FLO homologs from other systems. Detailed LFY expression through in situ hybridizations in the leaf and floral primordia would give an idea about the specific regions of its activity and thus help in elucidating its functions. The expression pattern of LFY in B. juncea was different from its closely related species A. thaliana and B. oleracea, though there was high level of sequence similarity amongst them. Isolation and characterization of the promoter and cis regulatory elements of the LFY gene from B. juncea may provide explanation for the variation in transcript profiles. The higher level of transcripts could be due to the presence of multiple copies of LFY gene in B. juncea. The analysis of the LFY promoter from B. juncea will also help in deciphering whether the transcriptional control of these regulators is direct or indirect and which proteins might act as mediators between these and the LFY gene.
4. ACKNOWLEDGEMENTS
S.D.R. was recipient of UGC fellowship, India (in the form of J.R.F. and S.R.F.). We are grateful to Prof. Shyam Prakash, I.A.R.I., New Delhi, India for providing Brassica juncea seeds and useful discussions, Prof. S. C. Maheswari and Dr. Nirmala Maheswari of I.C.G.E.B., New Delhi, India for critical comments.